Bymovirus reverse genetics: requirements for RNA2-encoded proteins in systemic infection
- PMID: 20447286
- PMCID: PMC6640240
- DOI: 10.1111/j.1364-3703.2010.00613.x
Bymovirus reverse genetics: requirements for RNA2-encoded proteins in systemic infection
Abstract
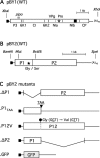
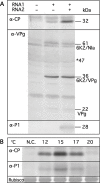
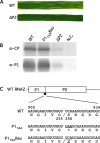
Barley yellow mosaic virus (BaYMV), the type species of the genus Bymovirus in the family Potyviridae in the picornavirus-like superfamily, causes a yellow mosaic disease of winter barley with significant yield losses in Europe and East Asia. Until now, infectious in vitro transcripts for the bipartite plus-sense RNA genome of any bymovirus species have not been available, rendering molecular analyses of bymovirus pathogenicity and the host resistance mechanisms difficult. In this study, we constructed the first cDNA clones of BaYMV RNA1 and RNA2, from which infectious RNA can be transcribed in vitro. Using in vitro transcripts, we showed that RNA1, which encodes eight proteins, including a viral proteinase NIa-Pro, the RNA-dependent RNA polymerase NIb, genome-linked viral protein VPg and the capsid protein CP, replicated autonomously in barley mesophyll protoplasts in the absence of RNA2 optimally at 15 degrees C, a temperature similar to the optimum for causing disease in barley fields. For systemic infection of barley plants, RNA1 alone was not sufficient and RNA2 was also required. Of the two proteins encoded on RNA2 (P1 with cysteine proteinase activity and P2 with unknown functions), P1 was essential and P2 was dispensable for systemic infectivity. The expression of both P1 and P2, but not the precursor polyprotein, together with RNA1 increased systemic infection and caused mosaic leaf symptoms. The infectious cDNA clones of BaYMV will be vital for future studies of bymovirus-host-vector interactions at the molecular level.
Figures
References
-
- Adams, M.J. , Swaby, A.G. and Jones, P. (1988) Confirmation of the transmission of barley yellow mosaic virus (BaYMV) by the fungus Polymyxa graminis . Ann. Appl. Biol. 112, 133–141.
-
- Adams, M.J. , Antoniw, F. and Mullins, J.G.L. (2001) Plant virus transmission by plasmodiophorid fungi is associated with distinctive transmembrane regions of virus‐encoded proteins. Arch. Virol. 146, 1139–1153. - PubMed
-
- Adams, M.J. , Antoniw, J.F. and Beaudoin, F. (2005) Overview and analysis of the polyprotein cleavage sites in the family Potyviridae . Mol. Plant Pathol. 6, 471–487. - PubMed
-
- Berger, P.H. , Adams, M.J. , Barnett, O.W. , Brunt, A.A. , Hammond, J. , Hill, J.H. , Jordan, R.L. , Kashiwazaki, S. , Rybicki, E. , Spence, N. , Stenger, D.C. , Ohki, S.T. , Uyeda, I. , Van Zaayen, A. , Valkonen, J. and Vetten, H. (2005) Family Potyviridae In: Virus Taxonomy. Eighth Report of the International Committee on Taxonomy of Viruses (Fauquet C.M., Mayo M.A., Maniloff J., Desselberger U. and Ball L.A., eds), pp. 819–841. San Diego, CA: Elsevier Academic Press.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

