Development of diagnostics in the search for an explanation of aerotoxic syndrome
- PMID: 20447373
- PMCID: PMC2900449
- DOI: 10.1016/j.ab.2010.04.032
Development of diagnostics in the search for an explanation of aerotoxic syndrome
Abstract
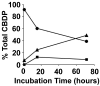
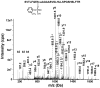
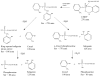
Aerotoxic syndrome is assumed to be caused by exposure to tricresyl phosphate, an additive in engine lubricants and hydraulic fluids that is activated to the toxic 2-(ortho-cresyl)-4H-1,3,2-benzodioxaphosphoran-2-one (CBDP). Currently, there is no laboratory evidence to support intoxication of airline crew members by CBDP. Our goal was to develop methods for testing in vivo exposure by identifying and characterizing biomarkers. Mass spectrometry was used to study the reaction of CBDP with human albumin, free tyrosine, and human butyrylcholinesterase. Human albumin made a covalent bond with CBDP, adding a mass of 170amu to Tyr411 to yield the o-cresyl phosphotyrosine derivative. Human butyrylcholinesterase made a covalent bond with CBDP on Ser198 to yield five adducts with added masses of 80, 108, 156, 170, and 186amu. The most abundant adduct had an added mass of 80amu from phosphate (HPO(3)), a surprising result given that no pesticide or nerve agent is known to yield phosphorylated serine with an added mass of 80amu. The next most abundant adduct had an added mass of 170amu to form o-cresyl phosphoserine. It is concluded that toxic gases or oil mists in cabin air may form adducts on plasma butyrylcholinesterase and albumin, detectable by mass spectrometry.
2010 Elsevier Inc. All rights reserved.
Figures
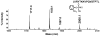

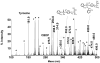
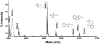

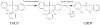
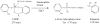
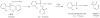
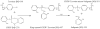
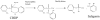
Similar articles
-
Reaction of cresyl saligenin phosphate, the organophosphorus agent implicated in aerotoxic syndrome, with human cholinesterases: mechanistic studies employing kinetics, mass spectrometry, and X-ray structure analysis.Chem Res Toxicol. 2011 Jun 20;24(6):797-808. doi: 10.1021/tx100447k. Epub 2011 Apr 18. Chem Res Toxicol. 2011. PMID: 21438623 Free PMC article.
-
Cresyl saligenin phosphate makes multiple adducts on free histidine, but does not form an adduct on histidine 438 of human butyrylcholinesterase.Chem Biol Interact. 2013 Mar 25;203(1):103-7. doi: 10.1016/j.cbi.2012.07.006. Epub 2012 Aug 8. Chem Biol Interact. 2013. PMID: 22898212 Free PMC article.
-
Cresyl saligenin phosphate, an organophosphorus toxicant, makes covalent adducts with histidine, lysine, and tyrosine residues of human serum albumin.Chem Res Toxicol. 2012 Aug 20;25(8):1752-61. doi: 10.1021/tx300215g. Epub 2012 Jul 26. Chem Res Toxicol. 2012. PMID: 22793878 Free PMC article.
-
The role of nanoparticles in bleed air in the etiology of Aerotoxic Syndrome: A review of cabin air-quality studies of 2003-2023.J Occup Environ Hyg. 2024 May-Jun;21(6):423-438. doi: 10.1080/15459624.2024.2327348. Epub 2024 Apr 9. J Occup Environ Hyg. 2024. PMID: 38593380 Review.
-
Analytical approaches for monitoring exposure to organophosphorus and carbamate agents through analysis of protein adducts.Drug Test Anal. 2012 Mar-Apr;4(3-4):246-61. doi: 10.1002/dta.1325. Epub 2012 Feb 22. Drug Test Anal. 2012. PMID: 22359362 Review.
Cited by
-
Exposure to tri-o-cresyl phosphate detected in jet airplane passengers.Toxicol Appl Pharmacol. 2011 Nov 1;256(3):337-47. doi: 10.1016/j.taap.2011.06.016. Epub 2011 Jun 24. Toxicol Appl Pharmacol. 2011. PMID: 21723309 Free PMC article.
-
Monoclonal Antibody That Recognizes Diethoxyphosphotyrosine-Modified Proteins and Peptides Independent of Surrounding Amino Acids.Chem Res Toxicol. 2017 Dec 18;30(12):2218-2228. doi: 10.1021/acs.chemrestox.7b00288. Epub 2017 Nov 28. Chem Res Toxicol. 2017. PMID: 29137457 Free PMC article.
-
Exposure to triaryl phosphates: metabolism and biomarkers of exposure.J Biol Phys Chem. 2011;11:10.4024/28FU11A.jbpc.11.04. doi: 10.4024/28FU11A.jbpc.11.04. J Biol Phys Chem. 2011. PMID: 24285929 Free PMC article.
-
Mortality from neurodegenerative diseases in a cohort of US flight attendants.Am J Ind Med. 2016 Jul;59(7):532-7. doi: 10.1002/ajim.22608. Epub 2016 May 17. Am J Ind Med. 2016. PMID: 27184412 Free PMC article.
-
Exploring adduct formation between human serum albumin and eleven organophosphate ester flame retardants and plasticizers using MALDI-TOF/TOF and LC-Q/TOF.Chemosphere. 2017 Aug;180:169-177. doi: 10.1016/j.chemosphere.2017.03.124. Epub 2017 Mar 31. Chemosphere. 2017. PMID: 28407546 Free PMC article.
References
-
- De Nola G, Kibby J, Mazurek W. Determination of ortho-cresyl phosphate isomers of tricresyl phosphate used in aircraft turbine engine oils by gas chromatography and mass spectrometry. J Chromatogr A. 2008;1200:211–6. - PubMed
-
- Winder C, Balouet JC. The toxicity of commercial jet oils. Environ Res. 2002;89:146–64. - PubMed
-
- van Netten C, Leung V. Hydraulic fluids and jet engine oil: pyrolysis and aircraft air quality. Arch Environ Health. 2001;56:181–6. - PubMed
-
- Henschler D. Tricresylphosphate poisoning; experimental clarification of problems of etiology and pathogenesis. Klin Wochenschr. 1958;36:663–74. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

