Factor Xa induces tissue factor expression in endothelial cells by P44/42 MAPK and NF-κB-dependent pathways
- PMID: 20451919
- PMCID: PMC2920361
- DOI: 10.1016/j.jss.2010.01.041
Factor Xa induces tissue factor expression in endothelial cells by P44/42 MAPK and NF-κB-dependent pathways
Abstract
Background: Tissue factor (TF) is an initiator of coagulation. The serine protease factor Xa (FXa) is the convergence point of the extrinsic and intrinsic components of the coagulation cascade. In addition to its hemostatic function, FXa elicits inflammatory responses in endothelial cells that may be important in surgical procedures in which inflammation is triggered. This study tested the hypothesis that FXa can up-regulate TF on vascular endothelial cells by a mitogen-activated protein kinase (MAPK)- and NF-κB-dependent pathway.
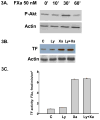
Methods and results: Incubation of cultured human umbilical vein endothelial cells (HUVECs) with FXa increased TF protein expression and activity in a dose-dependent manner. Pre-incubation of HUVECs with the serine protease inhibitor antithrombin, which targets not only thrombin but also FXa and FIXa, inhibited FXa-induced TF expression, but the selective thrombin inhibitor hirudin did not inhibit FXa-induced TF expression, ruling out a thrombin-mediated pathway. After 10 min incubation with HUVECs, FXa rapidly induced P44/42 MAPK activation (immunoblotting of phosphorylated P44/42 MAPK) with a peak at 30 min. The MEK 1/2 inhibitor PD98059 partially reduced FXa-induced TF expression and activity (3.82 ± 0.11 vs 6.54 ± 0.08 fmol/min/cm(2), P < 0.05). NF-κB was activated by FXa, confirmed by cytoplasmic IkBα degradation and increased NF-κB P65 nuclear translocation. Interruption of the NF-κB pathway by the IkBα phosphorylation inhibitor Bay 11-7802 abrogated FXa-induced TF protein expression and activity (1.93 ± 0.02 versus 6.54 ± 0.08 fmol/min/cm(2), P < 0.05). However, inhibition of PI3 kinase by LY 294002 did not attenuate FXa-induced TF protein expression and activity.
Conclusions: (1) FXa up-regulates TF protein expression and activity in HUVECs, (2) FXa-induced up-regulation of TF is independent of the thrombin-PAR1 pathway, and (3) the MAPK and NF-κB pathways, but not PI3 kinase pathway, are involved in FXa-induced TF expression on human umbilical endothelial cells. FXa may be a feed-forward alternative mechanism of activating TF expression and activity, thereby increasing a procoagulant state or inflammation. This mechanism may be important in the pro-inflammatory state initiated by cardiac surgical procedures.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures






Similar articles
-
Vascular endothelial growth factor production by fibroblasts in response to factor VIIa binding to tissue factor involves thrombin and factor Xa.Arterioscler Thromb Vasc Biol. 2000 May;20(5):1374-81. doi: 10.1161/01.atv.20.5.1374. Arterioscler Thromb Vasc Biol. 2000. PMID: 10807756
-
Oxidized phospholipids stimulate tissue factor expression in human endothelial cells via activation of ERK/EGR-1 and Ca(++)/NFAT.Blood. 2002 Jan 1;99(1):199-206. doi: 10.1182/blood.v99.1.199. Blood. 2002. PMID: 11756172
-
Macrophage migration inhibitory factor is induced by thrombin and factor Xa in endothelial cells.J Biol Chem. 2004 Apr 2;279(14):13729-37. doi: 10.1074/jbc.M400150200. Epub 2004 Jan 21. J Biol Chem. 2004. PMID: 14736878
-
Differential functions of tissue factor in the trans-activation of cellular signalling pathways.Atherosclerosis. 2007 Sep;194(1):88-101. doi: 10.1016/j.atherosclerosis.2006.10.010. Epub 2006 Nov 29. Atherosclerosis. 2007. PMID: 17137581
-
Synergistic effect of sphingosine 1-phosphate on thrombin-induced tissue factor expression in endothelial cells.Blood. 2003 Sep 1;102(5):1693-700. doi: 10.1182/blood-2002-11-3607. Epub 2003 May 1. Blood. 2003. PMID: 12730100
Cited by
-
Tissue factor, blood coagulation, and beyond: an overview.Int J Inflam. 2011;2011:367284. doi: 10.4061/2011/367284. Epub 2011 Sep 20. Int J Inflam. 2011. PMID: 21941675 Free PMC article.
-
Thrombin down-regulates tissue factor pathway inhibitor expression in a PI3K/nuclear factor-κB-dependent manner in human pleural mesothelial cells.Am J Respir Cell Mol Biol. 2015 Jun;52(6):674-82. doi: 10.1165/rcmb.2014-0084OC. Am J Respir Cell Mol Biol. 2015. PMID: 25303460 Free PMC article.
-
Circulating miR-320a-3p and miR-483-5p level associated with pharmacokinetic-pharmacodynamic profiles of rivaroxaban.Hum Genomics. 2022 Dec 28;16(1):72. doi: 10.1186/s40246-022-00445-5. Hum Genomics. 2022. PMID: 36578040 Free PMC article.
-
Prospective Application of Mesenchymal Stem Cell-Derived Exosomes in the Treatment of Disseminated Intravascular Coagulation.Int J Nanomedicine. 2024 Nov 16;19:11957-11971. doi: 10.2147/IJN.S467158. eCollection 2024. Int J Nanomedicine. 2024. PMID: 39569063 Free PMC article. Review.
-
The Intricate Role of Non-Coding RNAs in Sepsis-Associated Disseminated Intravascular Coagulation.Int J Mol Sci. 2023 Jan 30;24(3):2582. doi: 10.3390/ijms24032582. Int J Mol Sci. 2023. PMID: 36768905 Free PMC article. Review.
References
-
- Shimizu T, Nishihira J, Watanabe H, Abe R, Honda A, Ishibashi T, Shimizu H. Macrophage migration inhibitory factor is induced by thrombin and factor Xa in endothelial cells. J Biol Chem. 2004;279:13729–13737. - PubMed
-
- Bachli EB, Pech CM, Johnson KM, Johnson DJ, Tuddenham EGD, McVey JH. Factor Xa and thrombin, but not factor VIIa, elicit specific cellular responses in dermal fibroblasts. J Thromb Haemost. 2003;1:1935–1944. - PubMed
-
- Hollenstein UM, Pernerstorfer T, Homoncik M, Hansen JB, Finzen H, Handler S, Jilma B. Effect of factor X inhibition on coagulation activation and cytokine induction in human systemic inflammation. J Infect Disease. 2002;186:1270–1276. - PubMed
-
- Liu Y, Pelekanakis K, Woolkalis MJ. Thrombin and tumor necrosis factor α synergistically stimulate tissue factor expression in human endothelial cells. J Biol Chem. 2004;279:36142–36147. - PubMed
-
- Iba T, Kidokoro A, Fukunaga M, Fuse S, Suda M, Kunitada S, Hara T. Factor Xa-inhibitor (DX-9065a) modulates the leukocyte-endothelial cell interaction in endotoxemic rat. Shock. 2002;17:159–162. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

