Mammalian target of rapamycin signaling in the spinal cord is required for neuronal plasticity and behavioral hypersensitivity associated with neuropathy in the rat
- PMID: 20452291
- PMCID: PMC3000494
- DOI: 10.1016/j.jpain.2010.03.013
Mammalian target of rapamycin signaling in the spinal cord is required for neuronal plasticity and behavioral hypersensitivity associated with neuropathy in the rat
Abstract
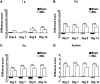
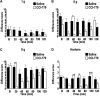
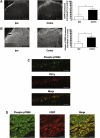
The protein kinase mammalian target of rapamycin (mTOR) regulates mRNA translation and is inhibited by rapamycin. Signaling pathways involving mTOR are implicated in physiological and pathophysiological processes. We determined the spinal effects of the rapamycin analogue cell cycle inhibitor (CCI)-779 on neuronal responses and behavioral hypersensitivity in a model of persistent neuropathic pain. We also assessed the anatomical distribution of spinal mTOR signaling pathways. Specifically, we ligated rat spinal nerves L5 and L6 to produce a model of neuropathic pain. After confirming neuropathy with behavioral testing, we obtained in vivo single-unit extracellular stimulus-evoked recordings from deep dorsal horn spinal neurons. We applied CCI-779 spinally in electrophysiological and behavioral studies and assessed its effects accordingly. We also used immunohistochemistry to probe for mTOR signaling pathways in dorsal root ganglia (DRG) and the spinal cord. We found that spinally administered CCI-779 rapidly attenuated calibrated mechanically but not thermally evoked neuronal responses and mechanically evoked behavioral responses. Immunohistochemistry showed presence of mTOR signaling pathways in nociceptive-specific C-fiber DRG and in neurons of inner lamina II of the spinal cord. We conclude that alterations in the activity of spinal mTOR signaling pathways are crucial to the full establishment of spinal neuronal plasticity and behavioral hypersensitivity associated with nerve injury.
Perspective: This study is consistent with growing evidence implicating mTOR signaling pathways as important modulators of persistent pain, providing novel insights into the molecular mechanisms of pain maintenance and potential for novel approaches into treating chronic pain.
Copyright © 2010 American Pain Society. Published by Elsevier Inc. All rights reserved.
Figures
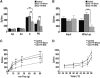

Similar articles
-
Formalin-induced behavioural hypersensitivity and neuronal hyperexcitability are mediated by rapid protein synthesis at the spinal level.Mol Pain. 2009 Jun 7;5:27. doi: 10.1186/1744-8069-5-27. Mol Pain. 2009. PMID: 19500426 Free PMC article.
-
mTOR and its downstream pathway are activated in the dorsal root ganglion and spinal cord after peripheral inflammation, but not after nerve injury.Brain Res. 2013 Jun 4;1513:17-25. doi: 10.1016/j.brainres.2013.04.003. Epub 2013 Apr 11. Brain Res. 2013. PMID: 23583278 Free PMC article.
-
Blocking Mammalian Target of Rapamycin (mTOR) Alleviates Neuropathic Pain Induced by Chemotherapeutic Bortezomib.Cell Physiol Biochem. 2018;48(1):54-62. doi: 10.1159/000491662. Epub 2018 Jul 11. Cell Physiol Biochem. 2018. Retraction in: Cell Physiol Biochem. 2022 Apr 30;56(2):234. doi: 10.33594/000000523. PMID: 29996117 Retracted.
-
Therapeutic potential of natural compounds from herbs and nutraceuticals in spinal cord injury: Regulation of the mTOR signaling pathway.Biomed Pharmacother. 2023 Jul;163:114905. doi: 10.1016/j.biopha.2023.114905. Epub 2023 May 17. Biomed Pharmacother. 2023. PMID: 37207430 Review.
-
Neuropathic pain: a maladaptive response of the nervous system to damage.Annu Rev Neurosci. 2009;32:1-32. doi: 10.1146/annurev.neuro.051508.135531. Annu Rev Neurosci. 2009. PMID: 19400724 Free PMC article. Review.
Cited by
-
mTOR, a new potential target for chronic pain and opioid-induced tolerance and hyperalgesia.Mol Pain. 2015 May 30;11:32. doi: 10.1186/s12990-015-0030-5. Mol Pain. 2015. PMID: 26024835 Free PMC article. Review.
-
Intrathecal rapamycin attenuates morphine-induced analgesic tolerance and hyperalgesia in rats with neuropathic pain.Transl Perioper Pain Med. 2015;2(2):27-34. Transl Perioper Pain Med. 2015. PMID: 26339682 Free PMC article.
-
Intact subepidermal nerve fibers mediate mechanical hypersensitivity via the activation of protein kinase C gamma in spared nerve injury.Mol Pain. 2016 Jun 13;12:1744806916656189. doi: 10.1177/1744806916656189. Print 2016. Mol Pain. 2016. PMID: 27296621 Free PMC article.
-
Targeting therapy-induced senescence as a novel strategy to combat chemotherapy-induced peripheral neuropathy.Support Care Cancer. 2024 Jan 5;32(1):85. doi: 10.1007/s00520-023-08287-0. Support Care Cancer. 2024. PMID: 38177894 Review.
-
Characterization of changes of pain behavior and signal transduction system in food-deprived mice.Anim Cells Syst (Seoul). 2018 Jun 28;22(4):227-233. doi: 10.1080/19768354.2018.1490348. eCollection 2018. Anim Cells Syst (Seoul). 2018. PMID: 30460102 Free PMC article.
References
-
- Attal N., Cruccu G., Haanpaa M., Hansson P., Jensen T.S., Nurmikko T., Sampaio C., Sindrup S., Wiffen P. EFNS guidelines on pharmacological treatment of neuropathic pain. Eur J Neurol. 2006;13:1153–1169. - PubMed
-
- Brouns I., Van Nassauw L., Van Genechten J., Majewski M., Scheuermann D.W., Timmermans J.P., Adriaensen D. Triple immunofluorescence staining with antibodies raised in the same species to study the complex innervation pattern of intrapulmonary chemoreceptors. J Histochem Cytochem. 2002;50:575–582. - PubMed
-
- Brown E.J., Schreiber S.L. A signaling pathway to translational control. Cell. 1996;86:517–520. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

