Signaling from axon guidance receptors
- PMID: 20452961
- PMCID: PMC2857166
- DOI: 10.1101/cshperspect.a001941
Signaling from axon guidance receptors
Abstract
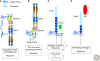
Determining how axon guidance receptors transmit signals to allow precise pathfinding decisions is fundamental to our understanding of nervous system development and may suggest new strategies to promote axon regeneration after injury or disease. Signaling mechanisms that act downstream of four prominent families of axon guidance cues--netrins, semaphorins, ephrins, and slits--have been extensively studied in both invertebrate and vertebrate model systems. Although details of these signaling mechanisms are still fragmentary and there appears to be considerable diversity in how different guidance receptors regulate the motility of the axonal growth cone, a number of common themes have emerged. Here, we review recent insights into how specific receptors for each of these guidance cues engage downstream regulators of the growth cone cytoskeleton to control axon guidance.
Figures
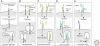


References
-
- Allen J, Chilton JK 2009. The specific targeting of guidance receptors within neurons: Who directs the directors? Dev Biol 327:4–11 - PubMed
-
- Beg AA, Sommer JE, Martin JH, Scheiffele P 2007. aα2-Chimaerin is an essential EphA4 effector in the assembly of neuronal locomotor circuits. Neuron 55:768–778 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
