Alpha-galactosidase A-Tat fusion enhances storage reduction in hearts and kidneys of Fabry mice
- PMID: 20454522
- PMCID: PMC2864812
- DOI: 10.2119/molmed.2009.00163
Alpha-galactosidase A-Tat fusion enhances storage reduction in hearts and kidneys of Fabry mice
Abstract
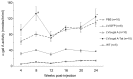
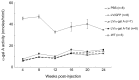
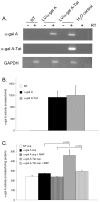
The protein transduction domain from human immunodeficiency virus (HIV) Tat allows proteins to penetrate the cell membrane. Enhanced cellular uptake of therapeutic proteins could benefit a number of disorders. This is especially true for lysosomal storage disorders (LSDs) where enzyme replacement therapy (ERT) and gene therapy have been developed. We developed a novel recombinant lentiviral vector (LV) that engineers expression of alpha-galactosidase A (alpha-gal A)-Tat fusion protein for correction of Fabry disease, the second-most prevalent LSD with manifestations in the brain, kidney and heart. In vitro experiments confirmed mannose-6-phosphate independent uptake of the fusion factor. Next, concentrated therapeutic LV was injected into neonatal Fabry mice. Analysis of tissues at 26 wks demonstrated similar alpha-gal A enzyme activities but enhanced globotriaosylceramide (Gb3) reduction in hearts and kidneys compared with the alpha-gal A LV control. This strategy might advance not only gene therapy for Fabry disease and other LSDs, but also ERT, especially for cardiac Fabry disease.
Figures

References
-
- Brady RO, et al. Enzymatic defect in Fabry’s disease. Ceramidetrihexosidase deficiency. N Engl J Med. 1967;276:1163–7. - PubMed
-
- Nakao S, et al. An atypical variant of Fabry’s disease in men with left ventricular hypertrophy. N Engl J Med. 1995;333:288–93. - PubMed
-
- Sachdev B, et al. Prevalence of Anderson-Fabry disease in male patients with late onset hypertrophic cardiomyopathy. Circulation. 2002;105:1407–11. - PubMed
-
- Banikazemi M, et al. Agalsidase-beta therapy for advanced Fabry disease: a randomized trial. Ann Intern Med. 2007;146:77–86. - PubMed
-
- Weidemann F, et al. Long-term effects of enzyme replacement therapy on Fabry cardiomyopathy: evidence for a better outcome with early treatment. Circulation. 2009;119:524–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

