Neural systems mediating seasonal breeding in the ewe
- PMID: 20456601
- PMCID: PMC2908208
- DOI: 10.1111/j.1365-2826.2010.02014.x
Neural systems mediating seasonal breeding in the ewe
Abstract
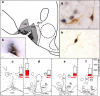
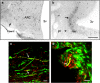
Seasonal reproduction in ewes is caused by a dramatic increase in response to oestradiol (E(2)) negative feedback during the nonbreeding (anoestrous) season. Considerable evidence supports the hypothesis that A15 dopaminergic neurones in the retrochiasmatic area (RCh) play a key role in these seasonal changes. These A15 neurones are stimulated by E(2) and inhibit gonadotrophin-releasing hormone (GnRH) secretion in anoestrus, but not the breeding season. Because A15 neurones do not contain oestrogen receptors-alpha (ER alpha), it is likely that E(2)-responsive afferents stimulate their activity when circulating E(2) levels increase during anoestrus. Retrograde tract tracing studies identified a limited set of ER alpha-containing afferents primarily found in four areas [ventromedial preoptic area, RCh, ventromedial and arcuate (ARC) nuclei]. Pharmacological and anatomical data are consistent with GABA- and glutamate-containing afferents controlling A15 activity in anoestrus, with E(2) inhibiting GABA and stimulating glutamate release at this time of year. Tract tracing demonstrated that A15 efferents project posteriorly to the median eminence and the ARC, suggesting possible direct actions on GnRH terminals or indirect actions via kisspeptin neurones in the ARC to inhibit GnRH in anoestrus. Identification of this neural circuitry sets the stage for the development of specific hypotheses for morphological or transmitter/receptor expression changes that would account for seasonal breeding in ewes.
Figures

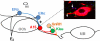
References
-
- Goodman RL, Inskeep EI. Neuroendocrine control of the ovarian cycle of the sheep. In: Neill JD, editor. Knobil and Neill's Physiology of Reproduction. 3rd ed Vol 2. Elsevier; Amsterdam, Netherlands: 2006. pp. 2389–2447.
-
- Malpaux B. Seasonal regulation of reproduction in mammals. Ibid. 2006;2:2231–2282.
-
- Karsch FJ, Bittman EL, Foster DL, Goodman RL, Legan SJ, Robinson JE. Neuroendocrine basis of seasonal reproduction. Recent Prog Horm Res. 1984;40:185–225. - PubMed
-
- Malpaux B, Daveau A, Maurice-Mandon F, Duarte G, Chemineau P. Evidence that melatonin acts in the premammillary hypothalamic area to control reproduction in the ewe: presence of binding sites and stimulation of luteinizing hormone secretion by in situ microimplant delivery. Endocrinology. 1998;139:1508–1516. - PubMed
-
- Goodman RL, Thiery JC, Malpaux B. The circannual rhythm controlling ovarian function in the ewe: possible neural elements. In: Touitou Y, editor. Biological clocks: Mechanisms and Applications Excerpta Med Intern Congress Ser Vol 1152. Elsevier Science BV; Amsterdam, Netherlands: 1998. pp. 363–371.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

