Induction and evasion of innate antiviral responses by hepatitis C virus
- PMID: 20457596
- PMCID: PMC2906263
- DOI: 10.1074/jbc.R109.099556
Induction and evasion of innate antiviral responses by hepatitis C virus
Abstract
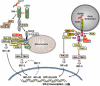
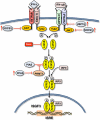
Persistent hepatitis C virus infection is associated with progressive hepatic fibrosis and liver cancer. Acute infection evokes several distinct innate immune responses, but these are partially or completely countered by the virus. Hepatitis C virus proteins serve dual functions in replication and immune evasion, acting to disrupt cellular signaling pathways leading to interferon synthesis, subvert Jak-STAT signaling to limit expression of interferon-stimulated genes, and block antiviral activities of interferon-stimulated genes. The net effect is a multilayered evasion of innate immunity, which negatively influences the subsequent development of antigen-specific adaptive immunity, thereby contributing to virus persistence and resistance to therapy.
Figures
Similar articles
-
Hepatitis A and hepatitis C viruses: divergent infection outcomes marked by similarities in induction and evasion of interferon responses.Semin Liver Dis. 2010 Nov;30(4):319-32. doi: 10.1055/s-0030-1267534. Epub 2010 Oct 19. Semin Liver Dis. 2010. PMID: 20960373 Review.
-
Insights into antiviral innate immunity revealed by studying hepatitis C virus.Cytokine. 2015 Aug;74(2):190-7. doi: 10.1016/j.cyto.2015.03.007. Epub 2015 Mar 25. Cytokine. 2015. PMID: 25819428 Free PMC article. Review.
-
Failure of innate and adaptive immune responses in controlling hepatitis C virus infection.FEMS Microbiol Rev. 2012 May;36(3):663-83. doi: 10.1111/j.1574-6976.2011.00319.x. Epub 2012 Jan 4. FEMS Microbiol Rev. 2012. PMID: 22142141 Review.
-
Multi-step regulation of interferon induction by hepatitis C virus.Arch Immunol Ther Exp (Warsz). 2013 Apr;61(2):127-38. doi: 10.1007/s00005-012-0214-x. Epub 2013 Jan 5. Arch Immunol Ther Exp (Warsz). 2013. PMID: 23292079 Review.
-
Activation and evasion of antiviral innate immunity by hepatitis C virus.J Mol Biol. 2014 Mar 20;426(6):1198-209. doi: 10.1016/j.jmb.2013.10.032. Epub 2013 Oct 31. J Mol Biol. 2014. PMID: 24184198 Free PMC article. Review.
Cited by
-
HCV infection selectively impairs type I but not type III IFN signaling.Am J Pathol. 2014 Jan;184(1):214-29. doi: 10.1016/j.ajpath.2013.10.005. Epub 2013 Nov 9. Am J Pathol. 2014. PMID: 24215913 Free PMC article.
-
Virus-specific mechanisms of carcinogenesis in hepatitis C virus associated liver cancer.Oncogene. 2011 Apr 28;30(17):1969-83. doi: 10.1038/onc.2010.594. Epub 2011 Jan 24. Oncogene. 2011. PMID: 21258404 Free PMC article. Review.
-
IL28B inhibits hepatitis C virus replication through the JAK-STAT pathway.J Hepatol. 2011 Aug;55(2):289-98. doi: 10.1016/j.jhep.2010.11.019. Epub 2010 Dec 13. J Hepatol. 2011. PMID: 21147189 Free PMC article.
-
Differential, type I interferon-mediated autophagic trafficking of hepatitis C virus proteins in mouse liver.Gastroenterology. 2011 Aug;141(2):674-85, 685.e1-6. doi: 10.1053/j.gastro.2011.04.060. Epub 2011 May 6. Gastroenterology. 2011. PMID: 21683701 Free PMC article.
-
STAT1 is Constitutively Activated in the T/C28a2 Immortalized Juvenile Human Chondrocyte Line and Stimulated by IL-6 Plus Soluble IL-6R.J Clin Cell Immunol. 2015 Apr;6(2):307. doi: 10.4172/2155-9899.1000307. J Clin Cell Immunol. 2015. PMID: 26213636 Free PMC article.
References
-
- Knipe D., Howley P., Griffin D. E., Martin M. A., Lamb R. A., Roizman B., Straus S. E. (2007) Fields Virology, 5th Ed., pp. 1253–1304, Lippincott Williams & Wilkins, Philadelphia
-
- Perz J. F., Armstrong G. L., Farrington L. A., Hutin Y. J., Bell B. P. (2006) J. Hepatol. 45, 529–538 - PubMed
-
- Pestka S. (2007) J. Biol. Chem. 282, 20047–20051 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

