A prominent role of the flagellin receptor FLAGELLIN-SENSING2 in mediating stomatal response to Pseudomonas syringae pv tomato DC3000 in Arabidopsis
- PMID: 20457804
- PMCID: PMC2899927
- DOI: 10.1104/pp.110.157016
A prominent role of the flagellin receptor FLAGELLIN-SENSING2 in mediating stomatal response to Pseudomonas syringae pv tomato DC3000 in Arabidopsis
Abstract
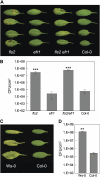
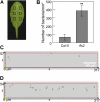
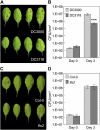
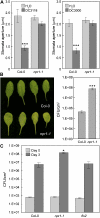
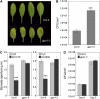
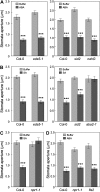
The FLAGELLIN-SENSING2 (FLS2) receptor kinase recognizes bacterial flagellin and initiates a battery of downstream defense responses to reduce bacterial invasion through stomata in the epidermis and bacterial multiplication in the apoplast of infected plants. Recent studies have shown that during Pseudomonas syringae pv tomato (Pst) DC3000 infection of Arabidopsis (Arabidopsis thaliana), FLS2-mediated immunity is actively suppressed by effector proteins (such as AvrPto and AvrPtoB) secreted through the bacterial type III secretion system (T3SS). We provide evidence here that T3SS effector-based suppression does not appear to be sufficient to overcome FLS2-based immunity during Pst DC3000 infection, but that the phytotoxin coronatine (COR) produced by Pst DC3000 also plays a critical role. COR-deficient mutants of Pst DC3000 are severely reduced in virulence when inoculated onto the leaf surface of wild-type Columbia-0 plants, but this defect was rescued almost fully in fls2 mutant plants. Although bacteria are thought to carry multiple microbe-associated molecular patterns, stomata of fls2 plants are completely unresponsive to COR-deficient mutant Pst DC3000 bacteria. The responses of fls2 plants were similar to those of the Arabidopsis G-protein alpha subunit1-3 mutant, which is defective in abscisic acid-regulated stomatal closure, but were distinct from those of the Arabidopsis non-expressor of PR genes1 mutant, which is defective in salicylic acid-dependent stomatal closure and apoplast defense. Epistasis analyses show that salicylic acid signaling acts upstream of abscisic acid signaling in bacterium-triggered stomatal closure. Taken together, these results suggest a particularly important role of FLS2-mediated resistance to COR-deficient mutant Pst DC3000 bacteria, and nonredundant roles of COR and T3SS effector proteins in the suppression of FLS2-mediated resistance in the Arabidopsis-Pst DC3000 interaction.
Figures
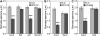
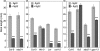
Similar articles
-
A genetic screen reveals Arabidopsis stomatal and/or apoplastic defenses against Pseudomonas syringae pv. tomato DC3000.PLoS Pathog. 2011 Oct;7(10):e1002291. doi: 10.1371/journal.ppat.1002291. Epub 2011 Oct 6. PLoS Pathog. 2011. PMID: 21998587 Free PMC article.
-
Positive regulation of BBX11 by NAC053 confers stomatal and apoplastic immunity against bacterial infection in Arabidopsis.New Phytol. 2025 May;246(4):1816-1833. doi: 10.1111/nph.70096. Epub 2025 Mar 20. New Phytol. 2025. PMID: 40110940
-
The Arabidopsis lectin receptor kinase LecRK-V.5 represses stomatal immunity induced by Pseudomonas syringae pv. tomato DC3000.PLoS Pathog. 2012 Feb;8(2):e1002513. doi: 10.1371/journal.ppat.1002513. Epub 2012 Feb 9. PLoS Pathog. 2012. PMID: 22346749 Free PMC article.
-
Pseudomonas syringae pv. tomato DC3000: a model pathogen for probing disease susceptibility and hormone signaling in plants.Annu Rev Phytopathol. 2013;51:473-98. doi: 10.1146/annurev-phyto-082712-102321. Epub 2013 May 31. Annu Rev Phytopathol. 2013. PMID: 23725467 Review.
-
Role of stomata in plant innate immunity and foliar bacterial diseases.Annu Rev Phytopathol. 2008;46:101-22. doi: 10.1146/annurev.phyto.121107.104959. Annu Rev Phytopathol. 2008. PMID: 18422426 Free PMC article. Review.
Cited by
-
Bacillus subtilis and Pseudomonas fluorescens Trigger Common and Distinct Systemic Immune Responses in Arabidopsis thaliana Depending on the Pathogen Lifestyle.Vaccines (Basel). 2020 Sep 4;8(3):503. doi: 10.3390/vaccines8030503. Vaccines (Basel). 2020. PMID: 32899695 Free PMC article.
-
Common and unique Arabidopsis proteins involved in stomatal susceptibility to Salmonella enterica and Pseudomonas syringae.FEMS Microbiol Lett. 2019 Aug 1;366(16):fnz197. doi: 10.1093/femsle/fnz197. FEMS Microbiol Lett. 2019. PMID: 31529017 Free PMC article.
-
Multi-Omics Revealed Molecular Mechanisms Underlying Guard Cell Systemic Acquired Resistance.Int J Mol Sci. 2020 Dec 27;22(1):191. doi: 10.3390/ijms22010191. Int J Mol Sci. 2020. PMID: 33375472 Free PMC article.
-
Ethylene signaling regulates accumulation of the FLS2 receptor and is required for the oxidative burst contributing to plant immunity.Plant Physiol. 2010 Sep;154(1):391-400. doi: 10.1104/pp.110.154567. Epub 2010 Jun 30. Plant Physiol. 2010. PMID: 20592040 Free PMC article.
-
AHL-priming functions via oxylipin and salicylic acid.Front Plant Sci. 2015 Jan 14;5:784. doi: 10.3389/fpls.2014.00784. eCollection 2014. Front Plant Sci. 2015. PMID: 25642235 Free PMC article. Review.
References
-
- Amborabe BE, Bonmort J, Fleurat-Lessard P, Roblin G. (2008) Early events induced by chitosan on plant cells. J Exp Bot 59: 2317–2324 - PubMed
-
- Bauer Z, Gómez-Gómez L, Boller T, Felix G. (2001) Sensitivity of different ecotypes and mutants of Arabidopsis thaliana toward the bacterial elicitor flagellin correlates with the presence of receptor-binding sites. J Biol Chem 276: 45669–45676 - PubMed
-
- Bender C, Rangaswamy V, Loper J. (1999) Polyketide production by plant associated pseudomonads. Annu Rev Phytopathol 37: 175–196 - PubMed
-
- Blanco F, Garreton V, Frey N, Dominguez C, Perez-Acle T, Van der Straeten D, Jordana X, Holuigue L. (2005) Identification of NPR1-dependent and independent genes early induced by salicylic acid treatment in Arabidopsis. Plant Mol Biol 59: 927–944 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

