PDZ protein mediated activity-dependent LTP/LTD developmental switch at rat retinocollicular synapses
- PMID: 20457829
- PMCID: PMC3774100
- DOI: 10.1152/ajpcell.00012.2010
PDZ protein mediated activity-dependent LTP/LTD developmental switch at rat retinocollicular synapses
Abstract
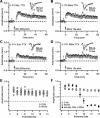
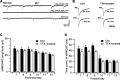
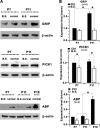
The insertion of amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors into the plasma membrane and removal via internalization are essential for regulating synaptic strength, which underlies the basic mechanism of learning and memory. The retinocollicular pathway undergoes synaptic refinement during development and shows a wide variety of long-term synaptic changes; however, still little is known about its underlying molecular regulation. Here we report a rapid developmental long-term potentiation (LTP)/long-term depression (LTD) switch and its intracellular mechanism at the rat retinocollicular pathway from postnatal day 5 (P5) to P14. Before P9, neurons always exhibited LTP, whereas LTD was observed only after P10. Blockade of GluR2/3-glutamate receptor-interacting protein (GRIP)/AMPA-receptor-binding protein (ABP)/protein interacting with C kinase 1 (PICK1) interactions with pep2-SVKI could sustain the LTP after P10. This suggests that the LTP/LTD switch relied on PDZ protein activities. Selective interruption of GluR2/3-PICK1 binding by pep2-EVKI blocked the long-lasting effects of both LTP and LTD, suggesting a role for PICK1 in the maintenance of long-term synaptic plasticity. Interestingly, synaptic expression of GRIP increased more than twofold from P7 to P11, whereas ABP and PICK1 expression levels remained stable. Blockade of spontaneous retinal input suppressed this increase and abolished the LTP/LTD switch. These results suggest that the increased GRIP synaptic expression may be a key regulatory factor in mediating the activity-dependent developmental LTP/LTD switch, whereas PICK1 may be required for both LTP and LTD to maintain their long-term effects.
Figures


Similar articles
-
An essential role for PICK1 in NMDA receptor-dependent bidirectional synaptic plasticity.Neuron. 2008 Mar 27;57(6):872-82. doi: 10.1016/j.neuron.2008.01.028. Neuron. 2008. PMID: 18367088 Free PMC article.
-
PDZ proteins interacting with C-terminal GluR2/3 are involved in a PKC-dependent regulation of AMPA receptors at hippocampal synapses.Neuron. 2000 Dec;28(3):873-86. doi: 10.1016/s0896-6273(00)00160-4. Neuron. 2000. PMID: 11163273
-
Properties of LTD and LTP of retinocollicular synaptic transmission in the developing rat superior colliculus.Eur J Neurosci. 2002 May;15(9):1421-32. doi: 10.1046/j.1460-9568.2002.01979.x. Eur J Neurosci. 2002. PMID: 12028352
-
Regulation of neuronal PKA signaling through AKAP targeting dynamics.Eur J Cell Biol. 2006 Jul;85(7):627-33. doi: 10.1016/j.ejcb.2006.01.010. Epub 2006 Feb 28. Eur J Cell Biol. 2006. PMID: 16504338 Review.
-
Modification of glutamate receptors by phospholipase A2: its role in adaptive neural plasticity.Cell Mol Life Sci. 2000 Oct;57(11):1542-50. doi: 10.1007/pl00000639. Cell Mol Life Sci. 2000. PMID: 11092449 Free PMC article. Review.
Cited by
-
PDZ and LIM Domain-Encoding Genes: Their Role in Cancer Development.Cancers (Basel). 2023 Oct 19;15(20):5042. doi: 10.3390/cancers15205042. Cancers (Basel). 2023. PMID: 37894409 Free PMC article. Review.
-
Glutamate receptor interacting protein acts within the prefrontal cortex to blunt cocaine seeking.Neuropharmacology. 2019 Oct;157:107672. doi: 10.1016/j.neuropharm.2019.107672. Epub 2019 Jun 21. Neuropharmacology. 2019. PMID: 31233823 Free PMC article.
-
Protein kinase C mediates hypoxia-induced long-term potentiation of NMDA neurotransmission in the visual retinocollicular pathway.Front Cell Neurosci. 2023 Feb 24;17:1141689. doi: 10.3389/fncel.2023.1141689. eCollection 2023. Front Cell Neurosci. 2023. PMID: 36909286 Free PMC article.
-
Disruption of glutamate receptor-interacting protein in nucleus accumbens enhances vulnerability to cocaine relapse.Neuropsychopharmacology. 2014 Feb;39(3):759-69. doi: 10.1038/npp.2013.265. Epub 2013 Oct 15. Neuropsychopharmacology. 2014. PMID: 24126453 Free PMC article.
References
-
- Barth AL, Malenka RC. NMDAR EPSC kinetics do not regulate the critical period for LTP at thalamocortical synapses. Nat Neurosci 4: 235–236, 2001 - PubMed
-
- Bartlett TE, Bannister NJ, Collett VJ, Dargan SL, Massey PV, Bortolotto ZA, Fitzjohn SM, Bashir ZI, Collingridge GL, Lodge D. Differential roles of NR2A and NR2B-containing NMDA receptors in LTP and LTD in the CA1 region of two-week old rat hippocampus. Neuropharmacology 52: 60–70, 2007 - PubMed
-
- Bear MF, Malenka RC. Synaptic plasticity: LTP and LTD. Curr Opin Neurobiol 4: 389–399, 1994 - PubMed
-
- Bear MF, Press WA, Connors BW. Long-term potentiation in slices of kitten visual cortex and the effects of NMDA receptor blockade. J Neurophysiol 67: 841–851, 1992 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

