A molecular mechanism underlying the neural-specific defect in torsinA mutant mice
- PMID: 20457914
- PMCID: PMC2906867
- DOI: 10.1073/pnas.0912877107
A molecular mechanism underlying the neural-specific defect in torsinA mutant mice
Abstract
A striking but poorly understood feature of many diseases is the unique involvement of neural tissue. One example is the CNS-specific disorder DYT1 dystonia, caused by a 3-bp deletion ("DeltaE") in the widely expressed gene TOR1A. Disease mutant knockin mice (Tor1a(DeltaE/DeltaE)) exhibit disrupted nuclear membranes selectively in neurons, mimicking the tissue specificity of the human disease and providing a model system in which to dissect the mechanisms underlying neural selectivity. Our in vivo studies demonstrate that lamina-associated polypeptide 1 (LAP1) and torsinB function with torsinA to maintain normal nuclear membrane morphology. Moreover, we show that nonneuronal cells express dramatically higher levels of torsinB and that RNAi-mediated depletion of torsinB (but not other torsin family members) causes nuclear membrane abnormalities in Tor1a(DeltaE/DeltaE) nonneuronal cells. The Tor1a(DeltaE/DeltaE) neural selective phenotype therefore arises because high levels of torsinB protect nonneuronal cells from the consequences of torsinA dysfunction, demonstrating how tissue specificity may result from differential susceptibility of cell types to insults that disrupt ubiquitous biological pathways.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
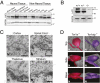
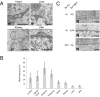
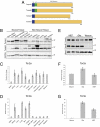
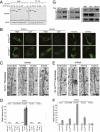
References
-
- Kustedjo K, Bracey MH, Cravatt BF. TorsinA and its torsion dystonia-associated mutant forms are lumenal glycoproteins that exhibit distinct subcellular localizations. J Biol Chem. 2000;275:27933–27939. - PubMed
-
- Ozelius LJ, et al. The early-onset torsion dystonia gene (DYT1) encodes an ATP-binding protein. Nat Genet. 1997;17:40–48. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

