Loss of leucine-rich repeat kinase 2 causes impairment of protein degradation pathways, accumulation of alpha-synuclein, and apoptotic cell death in aged mice
- PMID: 20457918
- PMCID: PMC2906862
- DOI: 10.1073/pnas.1004676107
Loss of leucine-rich repeat kinase 2 causes impairment of protein degradation pathways, accumulation of alpha-synuclein, and apoptotic cell death in aged mice
Abstract
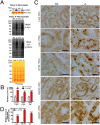
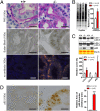
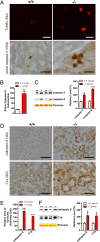
Mutations in leucine-rich repeat kinase 2 (LRRK2) are the most common genetic cause of Parkinson's disease. LRRK2 is a large protein containing a small GTPase domain and a kinase domain, but its physiological role is unknown. To identify the normal function of LRRK2 in vivo, we generated two independent lines of germ-line deletion mice. The dopaminergic system of LRRK2(-/-) mice appears normal, and numbers of dopaminergic neurons and levels of striatal dopamine are unchanged. However, LRRK2(-/-) kidneys, which suffer the greatest loss of LRRK compared with other organs, develop striking accumulation and aggregation of alpha-synuclein and ubiquitinated proteins at 20 months of age. The autophagy-lysosomal pathway is also impaired in the absence of LRRK2, as indicated by accumulation of lipofuscin granules as well as altered levels of LC3-II and p62. Furthermore, loss of LRRK2 dramatically increases apoptotic cell death, inflammatory responses, and oxidative damage. Collectively, our findings show that LRRK2 plays an essential and unexpected role in the regulation of protein homeostasis during aging, and suggest that LRRK2 mutations may cause Parkinson's disease and cell death via impairment of protein degradation pathways, leading to alpha-synuclein accumulation and aggregation over time.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Spillantini MG, et al. Alpha-synuclein in Lewy bodies. Nature. 1997;388:839–840. - PubMed
-
- Paisán-Ruíz C, et al. Cloning of the gene containing mutations that cause PARK8-linked Parkinson's disease. Neuron. 2004;44:595–600. - PubMed
-
- Zimprich A, et al. Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron. 2004;44:601–607. - PubMed
-
- Marín I. The Parkinson disease gene LRRK2: Evolutionary and structural insights. Mol Biol Evol. 2006;23:2423–2433. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

