Beneficial effect of aurothiomalate on murine malaria
- PMID: 20459650
- PMCID: PMC2875225
- DOI: 10.1186/1475-2875-9-118
Beneficial effect of aurothiomalate on murine malaria
Abstract
Background: Premature death of Plasmodium-infected erythrocytes is considered to favourably influence the clinical course of malaria. Aurothiomalate has previously been shown to trigger erythrocyte death or eryptosis, which is characterized by cell membrane scrambling leading to phosphatidylserine exposure at the cell surface. Phosphatidylserine-exposing cells are rapidly cleared from circulating blood. The present study thus tested whether sodium aurothiomalate influences the intraerythrocytic parasite development in vitro and the clinical course of murine malaria in vivo.
Methods: Human erythrocytes were infected with Plasmodium falciparum BinH in vitro and mice were infected (intraperitoneal injection of 1 x 106 parasitized murine erythrocytes) with Plasmodium berghei ANKA in vivo.
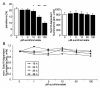
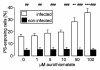
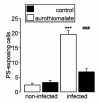
Results: Exposure to aurothiomalate significantly decreased the in vitro parasitemia of P. falciparum-infected human erythrocytes without influencing the intraerythrocytic DNA/RNA content. Administration of sodium aurothiomalate in vivo (daily 10 mg/kg b.w. s.c. from the 8th day of infection) enhanced the percentage of phosphatidylserine-exposing infected and noninfected erythrocytes in blood. All nontreated mice died within 30 days of infection. Aurothiomalate-treatment delayed the lethal course of malaria leading to survival of more than 50% of the mice 30 days after infection.
Conclusions: Sodium aurothiomalate influences the survival of Plasmodium berghei-infected mice, an effect only partially explained by stimulation of eryptosis.
Figures


References
-
- Kirk K. Membrane transport in the malaria-infected erythrocyte. Physiol Rev. 2001;81:495–537. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

