Preoperative CTLA-4 blockade: tolerability and immune monitoring in the setting of a presurgical clinical trial
- PMID: 20460488
- PMCID: PMC2919850
- DOI: 10.1158/1078-0432.CCR-10-0569
Preoperative CTLA-4 blockade: tolerability and immune monitoring in the setting of a presurgical clinical trial
Abstract
Purpose: Cytotoxic T lymphocyte associated antigen (CTLA-4) blockade is being explored in numerous clinical trials as an immune-based therapy for different malignancies. Our group conducted the first preoperative clinical trial with the anti-CTLA-4 antibody ipilimumab in 12 patients with localized urothelial carcinoma of the bladder.
Experimental design: Six patients were treated with 3 mg/kg/dose of anti-CTLA-4 and six patients were treated with 10 mg/kg/dose of antibody. Primary end points of the study were safety and immune monitoring.
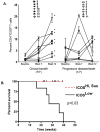
Results: Most drug-related adverse events consisted of grade 1/2 toxicities. All patients had measurable immunologic pharmacodynamic effects, consisting of an increased frequency of CD4+ICOShi T cells in tumor tissues and the systemic circulation. To determine if CD4+ICOShi T cells could be a correlative marker for clinical outcome after treatment with anti-CTLA-4, a cohort of metastatic melanoma patients was studied retrospectively for frequency of CD4+ICOShi T cells and survival. Data from this small cohort of patients indicated that an increased frequency of CD4+ICOShi T cells, sustained over a period of 12 weeks of therapy, correlates with increased likelihood of clinical benefit consisting of overall survival.
Conclusions: Our trial shows that anti-CTLA-4 therapy has a tolerable safety profile in the presurgical setting and that a preoperative model can be used to obtain biological data on human immune responses, which can efficiently guide the monitoring of patients treated in the metastatic disease setting.
Copyright (c) 2010 AACR.
Conflict of interest statement
Figures



Similar articles
-
CTLA-4 blockade increases IFNgamma-producing CD4+ICOShi cells to shift the ratio of effector to regulatory T cells in cancer patients.Proc Natl Acad Sci U S A. 2008 Sep 30;105(39):14987-92. doi: 10.1073/pnas.0806075105. Epub 2008 Sep 25. Proc Natl Acad Sci U S A. 2008. PMID: 18818309 Free PMC article. Clinical Trial.
-
CTLA-4 blockade enhances polyfunctional NY-ESO-1 specific T cell responses in metastatic melanoma patients with clinical benefit.Proc Natl Acad Sci U S A. 2008 Dec 23;105(51):20410-5. doi: 10.1073/pnas.0810114105. Epub 2008 Dec 12. Proc Natl Acad Sci U S A. 2008. PMID: 19074257 Free PMC article. Clinical Trial.
-
CD4 T cells require ICOS-mediated PI3K signaling to increase T-Bet expression in the setting of anti-CTLA-4 therapy.Cancer Immunol Res. 2014 Feb;2(2):167-76. doi: 10.1158/2326-6066.CIR-13-0155. Epub 2013 Nov 19. Cancer Immunol Res. 2014. PMID: 24778280 Free PMC article. Clinical Trial.
-
Anti-CTLA-4 antibody therapy: immune monitoring during clinical development of a novel immunotherapy.Semin Oncol. 2010 Oct;37(5):473-84. doi: 10.1053/j.seminoncol.2010.09.001. Semin Oncol. 2010. PMID: 21074063 Free PMC article. Review.
-
Emerging role of checkpoint inhibition in localized bladder cancer.Urol Oncol. 2016 Dec;34(12):548-555. doi: 10.1016/j.urolonc.2016.09.004. Epub 2016 Oct 21. Urol Oncol. 2016. PMID: 27776977 Review.
Cited by
-
Dysregulated NF-κB-Dependent ICOSL Expression in Human Dendritic Cell Vaccines Impairs T-cell Responses in Patients with Melanoma.Cancer Immunol Res. 2020 Dec;8(12):1554-1567. doi: 10.1158/2326-6066.CIR-20-0274. Epub 2020 Oct 13. Cancer Immunol Res. 2020. PMID: 33051240 Free PMC article. Clinical Trial.
-
ICOS+ Tregs: A Functional Subset of Tregs in Immune Diseases.Front Immunol. 2020 Aug 28;11:2104. doi: 10.3389/fimmu.2020.02104. eCollection 2020. Front Immunol. 2020. PMID: 32983168 Free PMC article. Review.
-
Immune Activation in Patients with Locally Advanced Cervical Cancer Treated with Ipilimumab Following Definitive Chemoradiation (GOG-9929).Clin Cancer Res. 2020 Nov 1;26(21):5621-5630. doi: 10.1158/1078-0432.CCR-20-0776. Epub 2020 Aug 18. Clin Cancer Res. 2020. PMID: 32816895 Free PMC article.
-
Prognostic value of immune checkpoint molecules in breast cancer.Biosci Rep. 2020 Jul 31;40(7):BSR20201054. doi: 10.1042/BSR20201054. Biosci Rep. 2020. PMID: 32602545 Free PMC article.
-
Advanced Urothelial Carcinoma: Overcoming Treatment Resistance through Novel Treatment Approaches.Front Pharmacol. 2013 Feb 6;4:3. doi: 10.3389/fphar.2013.00003. eCollection 2013. Front Pharmacol. 2013. PMID: 23390417 Free PMC article.
References
-
- Harding FA, McArthur JG, Gross JA, et al. CD28-mediated signalling co-stimulates murine T cells and prevents induction of anergy in T-cell clones. Nature. 1992;356:607–9. - PubMed
-
- Thompson CB, Allison JP. The emerging role of CTLA-4 as an immune attenuator. Immunity. 1997;7:445–50. - PubMed
-
- Leach DR, Krummel MF, Allison JP. Enhancement of antitumor immunity by CTLA-4 blockade. Science. 1996;271:1734–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

