When pathways collide: collaboration and connivance among signalling proteins in development
- PMID: 20461097
- PMCID: PMC4489880
- DOI: 10.1038/nrm2902
When pathways collide: collaboration and connivance among signalling proteins in development
Abstract
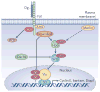
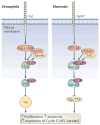
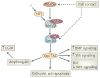
Signal transduction pathways interact at various levels to define tissue morphology, size and differentiation during development. Understanding the mechanisms by which these pathways collude has been greatly enhanced by recent insights into how shared components are independently regulated and how the activity of one system is contextualized by others. Traditionally, it has been assumed that the components of signalling pathways show pathway fidelity and act with a high degree of autonomy. However, as illustrated by the Wnt and Hippo pathways, there is increasing evidence that components are often shared between multiple pathways and other components talk to each other through multiple mechanisms.
Figures

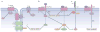
References
-
- Cohen P. The role of protein phosphorylation in neural and hormonal control of cellular activity. Nature. 1982;296:613–20. - PubMed
-
- van Amerongen R, Nusse R. Towards an integrated view of Wnt signaling in development. Development. 2009;136:3205–14. This reference and the one following (3) provide excellent and up to date reviews of Wnt signaling. - PubMed
-
- Zeng Q, Hong W. The emerging role of the hippo pathway in cell contact inhibition, organ size control, and cancer development in mammals. Cancer Cell. 2008;13:188–92. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

