Photorhabdus adhesion modification protein (Pam) binds extracellular polysaccharide and alters bacterial attachment
- PMID: 20462430
- PMCID: PMC2878306
- DOI: 10.1186/1471-2180-10-141
Photorhabdus adhesion modification protein (Pam) binds extracellular polysaccharide and alters bacterial attachment
Abstract
Background: Photorhabdus are Gram-negative nematode-symbiotic and insect-pathogenic bacteria. The species Photorhabdus asymbiotica is able to infect humans as well as insects. We investigated the secreted proteome of a clinical isolate of P. asymbiotica at different temperatures in order to identify proteins relevant to the infection of the two different hosts.
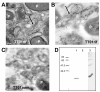
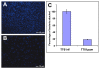
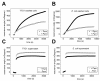
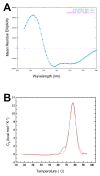
Results: A comparison of the proteins secreted by a clinical isolate of P. asymbiotica at simulated insect (28 degrees C) and human (37 degrees C) temperatures led to the identification of a small and highly abundant protein, designated Pam, that is only secreted at the lower temperature. The pam gene is present in all Photorhabdus strains tested and shows a high level of conservation across the whole genus, suggesting it is both ancestral to the genus and probably important to the biology of the bacterium. The Pam protein shows limited sequence similarity to the 13.6 kDa component of a binary toxin of Bacillus thuringiensis. Nevertheless, injection or feeding of heterologously produced Pam showed no insecticidal activity to either Galleria mellonella or Manduca sexta larvae. In bacterial colonies, Pam is associated with an extracellular polysaccharide (EPS)-like matrix, and modifies the ability of wild-type cells to attach to an artificial surface. Interestingly, Surface Plasmon Resonance (SPR) binding studies revealed that the Pam protein itself has adhesive properties. Although Pam is produced throughout insect infection, genetic knockout does not affect either insect virulence or the ability of P. luminescens to form a symbiotic association with its host nematode, Heterorhabditis bacteriophora.
Conclusions: We studied a highly abundant protein, Pam, which is secreted in a temperature-dependent manner in P. asymbiotica. Our findings indicate that Pam plays an important role in enhancing surface attachment in insect blood. Its association with exopolysaccharide suggests it may exert its effect through mediation of EPS properties. Despite its abundance and conservation in the genus, we find no evidence for a role of Pam in either virulence or symbiosis.
Figures



Similar articles
-
A genomic sample sequence of the entomopathogenic bacterium Photorhabdus luminescens W14: potential implications for virulence.Appl Environ Microbiol. 2000 Aug;66(8):3310-29. doi: 10.1128/AEM.66.8.3310-3329.2000. Appl Environ Microbiol. 2000. PMID: 10919786 Free PMC article.
-
The role of iron uptake in pathogenicity and symbiosis in Photorhabdus luminescens TT01.BMC Microbiol. 2010 Jun 22;10:177. doi: 10.1186/1471-2180-10-177. BMC Microbiol. 2010. PMID: 20569430 Free PMC article.
-
Photorhabdus luminescens genes induced upon insect infection.BMC Genomics. 2008 May 19;9:229. doi: 10.1186/1471-2164-9-229. BMC Genomics. 2008. PMID: 18489737 Free PMC article.
-
Comparative analysis of the Photorhabdus luminescens and the Yersinia enterocolitica genomes: uncovering candidate genes involved in insect pathogenicity.BMC Genomics. 2008 Jan 25;9:40. doi: 10.1186/1471-2164-9-40. BMC Genomics. 2008. PMID: 18221513 Free PMC article. Review.
-
The tc genes of Photorhabdus: a growing family.Trends Microbiol. 2001 Apr;9(4):185-91. doi: 10.1016/s0966-842x(01)01978-3. Trends Microbiol. 2001. PMID: 11286884 Review.
Cited by
-
Influence of the Photorhabdus luminescens phosphomannose isomerase gene, manA, on mannose utilization, exopolysaccharide structure, and biofilm formation.Appl Environ Microbiol. 2011 Feb;77(3):776-85. doi: 10.1128/AEM.02326-10. Epub 2010 Dec 10. Appl Environ Microbiol. 2011. PMID: 21148694 Free PMC article.
-
Dialkylresorcinols as bacterial signaling molecules.Proc Natl Acad Sci U S A. 2015 Jan 13;112(2):572-7. doi: 10.1073/pnas.1417685112. Epub 2014 Dec 30. Proc Natl Acad Sci U S A. 2015. PMID: 25550519 Free PMC article.
-
Organic Acid Exposure Enhances Virulence in Some Listeria monocytogenes Strains Using the Galleria mellonella Infection Model.Front Microbiol. 2021 Jul 6;12:675241. doi: 10.3389/fmicb.2021.675241. eCollection 2021. Front Microbiol. 2021. PMID: 34295317 Free PMC article.
-
From Insect to Man: Photorhabdus Sheds Light on the Emergence of Human Pathogenicity.PLoS One. 2015 Dec 17;10(12):e0144937. doi: 10.1371/journal.pone.0144937. eCollection 2015. PLoS One. 2015. PMID: 26681201 Free PMC article.
-
Structural and biophysical characterization of Bacillus thuringiensis insecticidal proteins Cry34Ab1 and Cry35Ab1.PLoS One. 2014 Nov 12;9(11):e112555. doi: 10.1371/journal.pone.0112555. eCollection 2014. PLoS One. 2014. PMID: 25390338 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

