Regulation of the creatine transporter by AMP-activated protein kinase in kidney epithelial cells
- PMID: 20462973
- PMCID: PMC2904179
- DOI: 10.1152/ajprenal.00162.2010
Regulation of the creatine transporter by AMP-activated protein kinase in kidney epithelial cells
Abstract
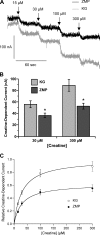
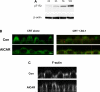
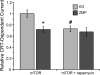
The metabolic sensor AMP-activated protein kinase (AMPK) regulates several transport proteins, potentially coupling transport activity to cellular stress and energy levels. The creatine transporter (CRT; SLC6A8) mediates creatine uptake into several cell types, including kidney epithelial cells, where it has been proposed that CRT is important for reclamation of filtered creatine, a process critical for total body creatine homeostasis. Creatine and phosphocreatine provide an intracellular, high-energy phosphate-buffering system essential for maintaining ATP supply in tissues with high energy demands. To test our hypothesis that CRT is regulated by AMPK in the kidney, we examined CRT and AMPK distribution in the kidney and the regulation of CRT by AMPK in cells. By immunofluorescence staining, we detected CRT at the apical pole in a polarized mouse S3 proximal tubule cell line and in native rat kidney proximal tubules, a distribution overlapping with AMPK. Two-electrode voltage-clamp (TEV) measurements of Na(+)-dependent creatine uptake into CRT-expressing Xenopus laevis oocytes demonstrated that AMPK inhibited CRT via a reduction in its Michaelis-Menten V(max) parameter. [(14)C]creatine uptake and apical surface biotinylation measurements in polarized S3 cells demonstrated parallel reductions in creatine influx and CRT apical membrane expression after AMPK activation with the AMP-mimetic compound 5-aminoimidazole-4-carboxamide-1-beta-D-ribofuranoside. In oocyte TEV experiments, rapamycin and the AMPK activator 5-aminoimidazole-4-carboxamide-1-beta-D-ribofuranosyl 5'-monophosphate (ZMP) inhibited CRT currents, but there was no additive inhibition of CRT by ZMP, suggesting that AMPK may inhibit CRT indirectly via the mammalian target of rapamycin pathway. We conclude that AMPK inhibits apical membrane CRT expression in kidney proximal tubule cells, which could be important in reducing cellular energy expenditure and unnecessary creatine reabsorption under conditions of local and whole body metabolic stress.
Figures




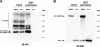
Similar articles
-
Inhibition of the KCa3.1 channels by AMP-activated protein kinase in human airway epithelial cells.Am J Physiol Cell Physiol. 2009 Feb;296(2):C285-95. doi: 10.1152/ajpcell.00418.2008. Epub 2008 Dec 3. Am J Physiol Cell Physiol. 2009. PMID: 19052260 Free PMC article.
-
Epithelial sodium channel inhibition by AMP-activated protein kinase in oocytes and polarized renal epithelial cells.J Biol Chem. 2005 May 6;280(18):17608-16. doi: 10.1074/jbc.M501770200. Epub 2005 Mar 7. J Biol Chem. 2005. PMID: 15753079
-
AMPK agonists ameliorate sodium and fluid transport and inflammation in cystic fibrosis airway epithelial cells.Am J Respir Cell Mol Biol. 2010 Jun;42(6):676-84. doi: 10.1165/2009-0147OC. Epub 2009 Jul 17. Am J Respir Cell Mol Biol. 2010. PMID: 19617399 Free PMC article.
-
Creatine biosynthesis and transport in health and disease.Biochimie. 2015 Dec;119:146-65. doi: 10.1016/j.biochi.2015.10.022. Epub 2015 Nov 2. Biochimie. 2015. PMID: 26542286 Review.
-
Beyond AICA riboside: in search of new specific AMP-activated protein kinase activators.IUBMB Life. 2009 Jan;61(1):18-26. doi: 10.1002/iub.135. IUBMB Life. 2009. PMID: 18798311 Free PMC article. Review.
Cited by
-
Activation of the metabolic sensor AMP-activated protein kinase inhibits aquaporin-2 function in kidney principal cells.Am J Physiol Renal Physiol. 2016 Nov 1;311(5):F890-F900. doi: 10.1152/ajprenal.00308.2016. Epub 2016 Aug 17. Am J Physiol Renal Physiol. 2016. PMID: 27534994 Free PMC article.
-
Accuracy of GFR estimation by the Cockroft and Gault, MDRD, and Wright equations in Oncology patients with renal impairment.Med Oncol. 2012 Jun;29(2):755-60. doi: 10.1007/s12032-011-9824-0. Epub 2011 Feb 1. Med Oncol. 2012. PMID: 21286862
-
Plasma creatine and incident type 2 diabetes in a general population-based cohort: The PREVEND study.Clin Endocrinol (Oxf). 2021 Apr;94(4):563-574. doi: 10.1111/cen.14396. Epub 2021 Jan 10. Clin Endocrinol (Oxf). 2021. PMID: 33348429 Free PMC article.
-
Cross-talk between AMP-activated protein kinase and renin-angiotensin system in uninephrectomised rats.J Renin Angiotensin Aldosterone Syst. 2016 Oct 26;17(4):1470320316673231. doi: 10.1177/1470320316673231. Print 2016 Oct. J Renin Angiotensin Aldosterone Syst. 2016. PMID: 27798124 Free PMC article.
-
Regulation of ion channels and transporters by AMP-activated kinase (AMPK).Channels (Austin). 2014;8(1):20-8. doi: 10.4161/chan.27423. Epub 2013 Dec 23. Channels (Austin). 2014. PMID: 24366036 Free PMC article. Review.
References
-
- Almaca J, Kongsuphol P, Hieke B, Ousingsawat J, Viollet B, Schreiber R, Amaral MD, Kunzelmann K. AMPK controls epithelial Na+ channels through Nedd4-2 and causes an epithelial phenotype when mutated. Pflügers Arch 458: 713–721, 2009 - PubMed
-
- Bens M, Vallet V, Cluzeaud F, Pascual-Letallec L, Kahn A, Rafestin-Oblin ME, Rossier BC, Vandewalle A. Corticosteroid-dependent sodium transport in a novel immortalized mouse collecting duct principal cell line. J Am Soc Nephrol 10: 923–934, 1999 - PubMed
-
- Bessman SP, Carpenter CL. The creatine-creatine phosphate energy shuttle. Annu Rev Biochem 54: 831–862, 1985 - PubMed
-
- Bhalla V, Oyster NM, Fitch AC, Wijngaarden MA, Neumann D, Schlattner U, Pearce D, Hallows KR. AMP-activated kinase inhibits the epithelial Na channel through functional regulation of the ubiquitin ligase Nedd4-2. J Biol Chem 281: 26159–26169, 2006 - PubMed
-
- Brown D, Lydon J, McLaughlin M, Stuart-Tilley A, Tyszkowski R, Alper S. Antigen retrieval in cryostat tissue sections and cultured cells by treatment with sodium dodecyl sulfate (SDS). Histochem Cell Biol 105: 261–267, 1996 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

