A spread-deficient cytomegalovirus for assessment of first-target cells in vaccination
- PMID: 20463067
- PMCID: PMC2897632
- DOI: 10.1128/JVI.02696-09
A spread-deficient cytomegalovirus for assessment of first-target cells in vaccination
Abstract
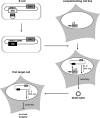
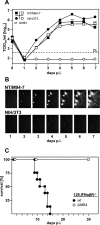
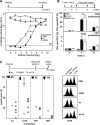
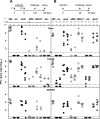
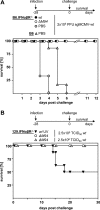
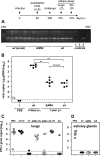
Human cytomegalovirus (HCMV) is a human pathogen that causes severe disease primarily in the immunocompromised or immunologically immature individual. To date, no vaccine is available. We describe use of a spread-deficient murine CMV (MCMV) as a novel approach for betaherpesvirus vaccination. To generate a spread-deficient MCMV, the conserved, essential gene M94 was deleted. Immunization with MCMV-DeltaM94 is apathogenic and protective against wild-type challenge even in highly susceptible IFNalphabetaR(-/-) mice. MCMV-DeltaM94 was able to induce a robust CD4(+) and CD8(+) T-cell response as well as a neutralizing antibody response comparable to that induced by wild-type infection. Endothelial cells were identified as activators of CD8(+) T cells in vivo. Thus, the vaccination with a spread-deficient betaherpesvirus is a safe and protective strategy and allows the linkage between cell tropism and immunogenicity. Furthermore, genomes of MCMV-DeltaM94 were present in lungs 12 months after infection, revealing first-target cells as sites of genome maintenance.
Figures

References
-
- Adler, S. P., S. E. Starr, S. A. Plotkin, S. H. Hempfling, J. Buis, M. L. Manning, and A. M. Best. 1995. Immunity induced by primary human cytomegalovirus infection protects against secondary infection among women of childbearing age. J. Infect. Dis. 171:26-32. - PubMed
-
- Andrews, D. M., C. E. Andoniou, F. Granucci, P. Ricciardi-Castagnoli, and M. A. Degli-Esposti. 2001. Infection of dendritic cells by murine cytomegalovirus induces functional paralysis. Nat. Immunol. 2:1077-1084. - PubMed
-
- Bale, J. F., Jr., S. J. Petheram, I. E. Souza, and J. R. Murph. 1996. Cytomegalovirus reinfection in young children. J. Pediatr. 128:347-352. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

