Opposing effects of a tyrosine-based sorting motif and a PDZ-binding motif regulate human T-lymphotropic virus type 1 envelope trafficking
- PMID: 20463077
- PMCID: PMC2898256
- DOI: 10.1128/JVI.01853-09
Opposing effects of a tyrosine-based sorting motif and a PDZ-binding motif regulate human T-lymphotropic virus type 1 envelope trafficking
Abstract
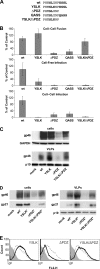
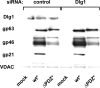
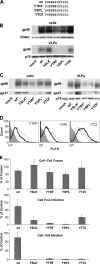
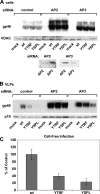
Human T-lymphotropic virus type 1 (HTLV-1) envelope (Env) glycoprotein mediates binding of the virus to its receptor on the surface of target cells and subsequent fusion of virus and cell membranes. To better understand the mechanisms that control HTLV-1 Env trafficking and activity, we have examined two protein-protein interaction motifs in the cytoplasmic domain of Env. One is the sequence YSLI, which matches the consensus YXXPhi motifs that are known to interact with various adaptor protein complexes; the other is the sequence ESSL at the C terminus of Env, which matches the consensus PDZ-binding motif. We show here that mutations that destroy the YXXPhi motif increased Env expression on the cell surface and increased cell-cell fusion activity. In contrast, mutation of the PDZ-binding motif greatly diminished Env expression in cells, which could be restored to wild-type levels either by mutating the YXXPhi motif or by silencing AP2 and AP3, suggesting that interactions with PDZ proteins oppose an Env degradation pathway mediated by AP2 and AP3. Silencing of the PDZ protein hDlg1 did not affect Env expression, suggesting that hDlg1 is not a binding partner for Env. Substitution of the YSLI sequence in HTLV-1 Env with YXXPhi elements from other cell or virus membrane-spanning proteins resulted in alterations in Env accumulation in cells, incorporation into virions, and virion infectivity. Env variants containing YXXPhi motifs that are predicted to have high-affinity interaction with AP2 accumulated to lower steady-state levels. Interestingly, mutations that destroy the YXXPhi motif resulted in viruses that were not infectious by cell-free or cell-associated routes of infection. Unlike YXXPhi, the function of the PDZ-binding motif manifests itself only in the producer cells; AP2 silencing restored the incorporation of PDZ-deficient Env into virus-like particles (VLPs) and the infectivity of these VLPs to wild-type levels.
Figures


Similar articles
-
Human Dlg protein binds to the envelope glycoproteins of human T-cell leukemia virus type 1 and regulates envelope mediated cell-cell fusion in T lymphocytes.J Cell Sci. 2004 Aug 1;117(Pt 17):3983-93. doi: 10.1242/jcs.01266. J Cell Sci. 2004. PMID: 15286176
-
Interactions of the cytoplasmic domains of human and simian retroviral transmembrane proteins with components of the clathrin adaptor complexes modulate intracellular and cell surface expression of envelope glycoproteins.J Virol. 1999 Feb;73(2):1350-61. doi: 10.1128/JVI.73.2.1350-1361.1999. J Virol. 1999. PMID: 9882340 Free PMC article.
-
Interaction of TSG101 with the PTAP Motif in Distinct Locations of Gag Determines the Incorporation of HTLV-1 Env into the Retroviral Virion.Int J Mol Sci. 2023 Nov 20;24(22):16520. doi: 10.3390/ijms242216520. Int J Mol Sci. 2023. PMID: 38003710 Free PMC article.
-
Molecular aspects of HTLV-1 entry: functional domains of the HTLV-1 surface subunit (SU) and their relationships to the entry receptors.Viruses. 2011 Jun;3(6):794-810. doi: 10.3390/v3060794. Epub 2011 Jun 15. Viruses. 2011. PMID: 21994754 Free PMC article. Review.
-
Molecular mechanisms affecting HTLV type 1-dependent fusion at the cell membrane: implications for inhibiting viral transmission.AIDS Res Hum Retroviruses. 2000 Nov 1;16(16):1731-6. doi: 10.1089/08892220050193227. AIDS Res Hum Retroviruses. 2000. PMID: 11080818 Review.
Cited by
-
Retroviral env glycoprotein trafficking and incorporation into virions.Mol Biol Int. 2012;2012:682850. doi: 10.1155/2012/682850. Epub 2012 Jul 2. Mol Biol Int. 2012. PMID: 22811910 Free PMC article.
-
Internalization of rabies virus glycoprotein differs between pathogenic and attenuated virus strains.J Gen Virol. 2023 Dec;104(12):001935. doi: 10.1099/jgv.0.001935. J Gen Virol. 2023. PMID: 38063294 Free PMC article.
-
Emerging theme: cellular PDZ proteins as common targets of pathogenic viruses.J Virol. 2011 Nov;85(22):11544-56. doi: 10.1128/JVI.05410-11. Epub 2011 Jul 20. J Virol. 2011. PMID: 21775458 Free PMC article. Review.
-
Molecular determinants of human T-lymphotropic virus type 1 transmission and spread.Viruses. 2011 Jul;3(7):1131-65. doi: 10.3390/v3071131. Epub 2011 Jul 12. Viruses. 2011. PMID: 21994774 Free PMC article. Review.
-
Stability of the HTLV-1 glycoprotein 46 (gp46) gene in an endemic region of the Brazilian Amazon and the presence of a significant mutation (N93D) in symptomatic patients.Virol J. 2018 May 2;15(1):80. doi: 10.1186/s12985-018-0984-9. Virol J. 2018. PMID: 29716616 Free PMC article.
References
-
- Berlioz-Torrent, C., B. L. Shacklett, L. Erdtmann, L. Delamarre, I. Bouchaert, P. Sonigo, M. C. Dokhelar, and R. Benarous. 1999. Interactions of the cytoplasmic domains of human and simian retroviral transmembrane proteins with components of the clathrin adaptor complexes modulate intracellular and cell surface expression of envelope glycoproteins. J. Virol. 73:1350-1361. - PMC - PubMed
-
- Blot, V., L. Delamarre, F. Perugi, D. Pham, S. Benichou, R. Benarous, T. Hanada, A. H. Chishti, M. C. Dokhelar, and C. Pique. 2004. Human Dlg protein binds to the envelope glycoproteins of human T-cell leukemia virus type 1 and regulates envelope mediated cell-cell fusion in T lymphocytes. J. Cell Sci. 117:3983-3993. - PubMed
-
- Bobkova, M., J. Stitz, M. Engelstadter, K. Cichutek, and C. J. Buchholz. 2002. Identification of R-peptides in envelope proteins of C-type retroviruses. J. Gen. Virol. 83:2241-2246. - PubMed
-
- Boge, M., S. Wyss, J. S. Bonifacino, and M. Thali. 1998. A membrane-proximal tyrosine-based signal mediates internalization of the HIV-1 envelope glycoprotein via interaction with the AP-2 clathrin adaptor. J. Biol. Chem. 273:15773-15778. - PubMed
-
- Bonifacino, J. S., and L. M. Traub. 2003. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 72:395-447. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

