Sodium/calcium exchangers selectively regulate calcium signaling in mouse taste receptor cells
- PMID: 20463203
- PMCID: PMC2904227
- DOI: 10.1152/jn.00118.2010
Sodium/calcium exchangers selectively regulate calcium signaling in mouse taste receptor cells
Abstract
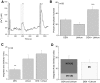
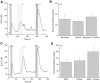
Taste cells use multiple signaling mechanisms to generate appropriate cellular responses to discrete taste stimuli. Some taste stimuli activate G protein coupled receptors (GPCRs) that cause calcium release from intracellular stores while other stimuli depolarize taste cells to cause calcium influx through voltage-gated calcium channels (VGCCs). While the signaling mechanisms that initiate calcium signals have been described in taste cells, the calcium clearance mechanisms (CCMs) that contribute to the termination of these signals have not been identified. In this study, we used calcium imaging to define the role of sodium-calcium exchangers (NCXs) in the termination of evoked calcium responses. We found that NCXs regulate the calcium signals that rely on calcium influx at the plasma membrane but do not significantly contribute to the calcium signals that depend on calcium release from internal stores. Our data indicate that this selective regulation of calcium signals by NCXs is due primarily to their location in the cell rather than to the differences in cytosolic calcium loads. This is the first report to define the physiological role for any of the CCMs utilized by taste cells to regulate their evoked calcium responses.
Figures


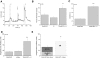
Similar articles
-
Calcium signaling in taste cells.Biochim Biophys Acta. 2015 Sep;1853(9):2025-32. doi: 10.1016/j.bbamcr.2014.11.013. Epub 2014 Nov 16. Biochim Biophys Acta. 2015. PMID: 25450977 Free PMC article. Review.
-
Sodium-calcium exchangers contribute to the regulation of cytosolic calcium levels in mouse taste cells.J Physiol. 2009 Aug 15;587(Pt 16):4077-89. doi: 10.1113/jphysiol.2009.173567. Epub 2009 Jul 6. J Physiol. 2009. PMID: 19581381 Free PMC article.
-
Calcium signaling in taste cells: regulation required.Chem Senses. 2010 Nov;35(9):753-65. doi: 10.1093/chemse/bjq082. Epub 2010 Aug 25. Chem Senses. 2010. PMID: 20739430 Free PMC article. Review.
-
Ryanodine receptors selectively contribute to the formation of taste-evoked calcium signals in mouse taste cells.Eur J Neurosci. 2010 Dec;32(11):1825-35. doi: 10.1111/j.1460-9568.2010.07463.x. Epub 2010 Oct 19. Eur J Neurosci. 2010. PMID: 20955474 Free PMC article.
-
Mitochondrial calcium buffering contributes to the maintenance of Basal calcium levels in mouse taste cells.J Neurophysiol. 2008 Oct;100(4):2177-91. doi: 10.1152/jn.90534.2008. Epub 2008 Aug 6. J Neurophysiol. 2008. PMID: 18684902 Free PMC article.
Cited by
-
A subset of broadly responsive Type III taste cells contribute to the detection of bitter, sweet and umami stimuli.PLoS Genet. 2020 Aug 13;16(8):e1008925. doi: 10.1371/journal.pgen.1008925. eCollection 2020 Aug. PLoS Genet. 2020. PMID: 32790785 Free PMC article.
-
AP1 transcription factors are required to maintain the peripheral taste system.Cell Death Dis. 2016 Oct 27;7(10):e2433. doi: 10.1038/cddis.2016.343. Cell Death Dis. 2016. PMID: 27787515 Free PMC article.
-
Calcium signaling in taste cells.Biochim Biophys Acta. 2015 Sep;1853(9):2025-32. doi: 10.1016/j.bbamcr.2014.11.013. Epub 2014 Nov 16. Biochim Biophys Acta. 2015. PMID: 25450977 Free PMC article. Review.
-
Defining the role of TRPM4 in broadly responsive taste receptor cells.Front Cell Neurosci. 2023 Mar 22;17:1148995. doi: 10.3389/fncel.2023.1148995. eCollection 2023. Front Cell Neurosci. 2023. PMID: 37032837 Free PMC article.
-
The WT1-BASP1 complex is required to maintain the differentiated state of taste receptor cells.Life Sci Alliance. 2019 Jun 5;2(3):e201800287. doi: 10.26508/lsa.201800287. Print 2019 Jun. Life Sci Alliance. 2019. PMID: 31167803 Free PMC article.
References
-
- Akabas MH, Dodd J, Al-Awqati Q. A bitter substance induces a rise in intracellular calcium in a subpopulation of rat taste cells. Science 242: 1047–1050, 1988 - PubMed
-
- Augustine GJ, Santamaria F, Tanaka K. Local calcium signaling in neurons. Neuron 40: 331–346, 2003 - PubMed
-
- Berridge MJ, Bootman MD, Lipp P. Calcium—a life and death signal. Nature 395: 645–648, 1998 - PubMed
-
- Blaustein MP. Calcium transport and buffering in neurons. Trends Neurosci 11: 438–443, 1988 - PubMed
-
- Blaustein MP, Juhaszova M, Golovina VA, Church PJ, Stanley EF. Na/Ca exchanger and PMCA localization in neurons and astrocytes: functional implications. Ann NY Acad Sci 976: 356–366, 2002 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

