Muscarinic signaling in the cochlea: presynaptic and postsynaptic effects on efferent feedback and afferent excitability
- PMID: 20463237
- PMCID: PMC3332094
- DOI: 10.1523/JNEUROSCI.5080-09.2010
Muscarinic signaling in the cochlea: presynaptic and postsynaptic effects on efferent feedback and afferent excitability
Abstract
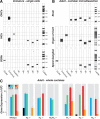
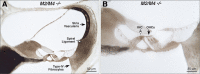
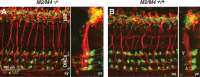
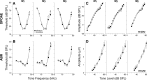
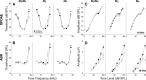
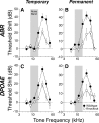
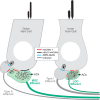
Acetylcholine is the major neurotransmitter of the olivocochlear efferent system, which provides feedback to cochlear hair cells and sensory neurons. To study the role of cochlear muscarinic receptors, we studied receptor localization with immunohistochemistry and reverse transcription-PCR and measured olivocochlear function, cochlear responses, and histopathology in mice with targeted deletion of each of the five receptor subtypes. M2, M4, and M5 were detected in microdissected immature (postnatal days 10-13) inner hair cells and spiral ganglion cells but not outer hair cells. In the adult (6 weeks), the same transcripts were found in microdissected organ of Corti and spiral ganglion samples. M2 protein was found, by immunohistochemistry, in olivocochlear fibers in both outer and inner hair cell areas. M3 mRNA was amplified only from whole cochleas, and M1 message was never seen in wild-type ears. Auditory brainstem responses (ABRs) and distortion product otoacoustic emissions (DPOAEs) were unaffected by loss of Gq-coupled receptors (M1, M3, or M5), as were shock-evoked olivocochlear effects and vulnerability to acoustic injury. In contrast, loss of Gi-coupled receptors (M2 and/or M4) decreased neural responses without affecting DPOAEs (at low frequencies). This phenotype and the expression pattern are consistent with excitatory muscarinic signaling in cochlear sensory neurons. At high frequencies, both ABRs and DPOAEs were attenuated by loss of M2 and/or M4, and the vulnerability to acoustic injury was dramatically decreased. This aspect of the phenotype and the expression pattern are consistent with a presynaptic role for muscarinic autoreceptors in decreasing ACh release from olivocochlear terminals during high-level acoustic stimulation and suggest that muscarinic antagonists could enhance the resistance of the inner ear to noise-induced hearing loss.
Figures

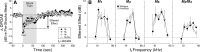
References
-
- Bartolami S, Ripoll C, Planche M, Pujol R. Localization of functional muscarinic receptors in the rat cochlea: evidence for efferent presynaptic autoreceptors. Brain Res. 1993;626:200–209. - PubMed
-
- Brown DA, Sihra TS. Presynaptic signaling by heterotrimeric G-proteins. Handb Exp Pharmacol. 2008;184:207–260. - PubMed
-
- Caulfield MP. Muscarinic receptors-characterization, coupling and function. Pharmacol Ther. 1993;58:319–379. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
