Electrochemical growth behavior, surface properties, and enhanced in vivo bone response of TiO2 nanotubes on microstructured surfaces of blasted, screw-shaped titanium implants
- PMID: 20463928
- PMCID: PMC2865007
- DOI: 10.2147/ijn.s8012
Electrochemical growth behavior, surface properties, and enhanced in vivo bone response of TiO2 nanotubes on microstructured surfaces of blasted, screw-shaped titanium implants
Abstract
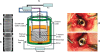
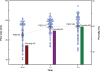
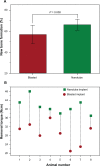
TiO(2) nanotubes are fabricated on TiO(2) grit-blasted, screw-shaped rough titanium (ASTM grade 4) implants (3.75 x 7 mm) using potentiostatic anodization at 20 V in 1 M H(3)PO(4) + 0.4 wt.% HF. The growth behavior and surface properties of the nanotubes are investigated as a function of the reaction time. The results show that vertically aligned nanotubes of approximately 700 nm in length, with highly ordered structures of approximately 40 nm spacing and approximately 15 nm wall thickness may be grown independent of reaction time. The geometrical properties of nanotubes increase with reaction time (mean pore size, pore size distribution [PSD], and porosity approximately 90 nm, approximately 40-127 nm and 45%, respectively for 30 minutes; approximately 107 nm, approximately 63-140 nm and 56% for one hour; approximately 108 nm, approximately 58-150 nm and 60% for three hours). It is found that the fluorinated chemistry of the nanotubes of F-TiO(2), TiOF(2), and F-Ti-O with F ion incorporation of approximately 5 at.%, and their amorphous structure is the same regardless of the reaction time, while the average roughness (Sa) gradually decreases and the developed surface area (Sdr) slightly increases with reaction time. The results of studies on animals show that, despite their low roughness values, after six weeks the fluorinated TiO(2) nanotube implants in rabbit femurs demonstrate significantly increased osseointegration strengths (41 vs 29 Ncm; P = 0.008) and new bone formation (57.5% vs 65.5%; P = 0.008) (n = 8), and reveal more frequently direct bone/cell contact at the bone-implant interface by high-resolution scanning electron microscope observations as compared with the blasted, moderately rough implants that have hitherto been widely used for clinically favorable performance. The results of the animal studies constitute significant evidence that the presence of the nanotubes and the resulting fluorinated surface chemistry determine the nature of the bone responses to the implants. The present in vivo results point to potential applications of the TiO(2) nanotubes in the field of bone implants and bone tissue engineering.
Keywords: electrochemical fabrication; fluorinated TiO2 nanotubes; in vivo bone response; osseointegrated titanium implant; surface properties.
Figures






References
-
- Zwilling V, Aucouturier M, Darque-Ceretti E. Anodic oxidation of titanium and TA6V alloy in chromic media. An electrochemical approach. Electrochimi Acta. 1999;45(6):921–929.
-
- Ghicov A, Schmuki P. Self-ordering electrochemistry: a review on growth and functionality of TiO2 nanotubes and other self-aligned MO(x) structures. Chem Commun. 2009;(20):2791–2808. - PubMed
-
- Macak JM, Tsuchiya H, Ghicov A, et al. TiO2 nanotubes: Self-organized electrochemical formation, properties and applications. Curr Opin Solid State Mater Sci. 2007;11(1–2):3–18.
-
- Mor GK, Varghese OK, Paulose M, Shankar K, Grimes CA. A review on highly ordered, vertically oriented TiO2 nanotube arrays: Fabrication, material properties, and solar energy applications. Sol Energy Mater Sol Cells. 2006;90(14):2011–2075.
-
- Chen X, Mao SS. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem Rev. 2007;107(7):2891–2959. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

