De novo constitutional MLH1 epimutations confer early-onset colorectal cancer in two new sporadic Lynch syndrome cases, with derivation of the epimutation on the paternal allele in one
- PMID: 20473912
- PMCID: PMC3794437
- DOI: 10.1002/ijc.25422
De novo constitutional MLH1 epimutations confer early-onset colorectal cancer in two new sporadic Lynch syndrome cases, with derivation of the epimutation on the paternal allele in one
Abstract
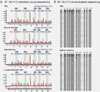
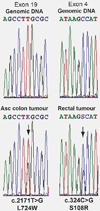
Lynch syndrome is an autosomal dominant cancer predisposition syndrome classically caused by germline mutations of the mismatch repair genes, MLH1, MSH2, MSH6 and PMS2. Constitutional epimutations of the MLH1 gene, characterized by soma-wide methylation of a single allele of the promoter and allelic transcriptional silencing, have been identified in a subset of Lynch syndrome cases lacking a sequence mutation in MLH1. We report two individuals with no family history of colorectal cancer who developed that disease at age 18 and 20 years. In both cases, cancer had arisen because of the de novo occurrence of a constitutional MLH1 epimutation and somatic loss-of-heterozygosity of the functional allele in the tumors. We show for the first time that the epimutation in one case arose on the paternally inherited allele. Analysis of 13 tumors from seven individuals with constitutional MLH1 epimutations showed eight tumors had lost the second MLH1 allele, two tumors had a novel pathogenic missense mutation and three had retained heterozygosity. Only 1 of 12 tumors demonstrated the BRAF V600E mutation and 3 of 11 tumors harbored a mutation in KRAS. The finding that epimutations can originate on the paternal allele provides important new insights into the mechanism of origin of epimutations. It is clear that the second hit in MLH1 epimutation-associated tumors typically has a genetic not epigenetic basis. Individuals with mismatch repair-deficient cancers without the BRAF V600E mutation are candidates for germline screening for sequence or methylation changes in MLH1.
Copyright © 2010 UICC.
Figures


Similar articles
-
Finding the needle in a haystack: identification of cases of Lynch syndrome with MLH1 epimutation.Fam Cancer. 2016 Jul;15(3):413-22. doi: 10.1007/s10689-016-9887-3. Fam Cancer. 2016. PMID: 26886015 Review.
-
Evidence of constitutional MLH1 epimutation associated to transgenerational inheritance of cancer susceptibility.Hum Mutat. 2012 Jan;33(1):180-8. doi: 10.1002/humu.21617. Epub 2011 Oct 31. Hum Mutat. 2012. PMID: 21953887
-
Constitutional (germline) MLH1 epimutation as an aetiological mechanism for hereditary non-polyposis colorectal cancer.J Med Genet. 2009 Dec;46(12):793-802. doi: 10.1136/jmg.2009.068122. Epub 2009 Jun 29. J Med Genet. 2009. PMID: 19564652 Review.
-
MLH1 methylation screening is effective in identifying epimutation carriers.Eur J Hum Genet. 2012 Dec;20(12):1256-64. doi: 10.1038/ejhg.2012.136. Epub 2012 Jul 4. Eur J Hum Genet. 2012. PMID: 22763379 Free PMC article.
-
Detection of allelic imbalance in MLH1 expression by pyrosequencing serves as a tool for the identification of germline defects in Lynch syndrome.Fam Cancer. 2010 Sep;9(3):345-56. doi: 10.1007/s10689-009-9314-0. Fam Cancer. 2010. PMID: 20063070
Cited by
-
Lynch syndrome associated with two MLH1 promoter variants and allelic imbalance of MLH1 expression.Hum Mutat. 2015 Jun;36(6):622-30. doi: 10.1002/humu.22785. Epub 2015 Apr 17. Hum Mutat. 2015. PMID: 25762362 Free PMC article.
-
Constitutional Mosaic Epimutations - a hidden cause of cancer?Cell Stress. 2019 Mar 22;3(4):118-135. doi: 10.15698/cst2019.04.183. Cell Stress. 2019. PMID: 31225507 Free PMC article. Review.
-
Finding the needle in a haystack: identification of cases of Lynch syndrome with MLH1 epimutation.Fam Cancer. 2016 Jul;15(3):413-22. doi: 10.1007/s10689-016-9887-3. Fam Cancer. 2016. PMID: 26886015 Review.
-
Towards the development of next-generation lung cancer immunotherapy.Transl Lung Cancer Res. 2025 Jun 30;14(6):2257-2271. doi: 10.21037/tlcr-2024-1097. Epub 2025 Jun 25. Transl Lung Cancer Res. 2025. PMID: 40673103 Free PMC article. Review.
-
MSH3-deficiency initiates EMAST without oncogenic transformation of human colon epithelial cells.PLoS One. 2012;7(11):e50541. doi: 10.1371/journal.pone.0050541. Epub 2012 Nov 27. PLoS One. 2012. PMID: 23209772 Free PMC article.
References
-
- Peltomaki P. Lynch syndrome genes. Fam Cancer. 2005;4:227–232. - PubMed
-
- Taylor CF, Charlton RS, Burn J, Sheridan E, Taylor GR. Genomic deletions in MSH2 or MLH1 are a frequent cause of hereditary non-polyposis colorectal cancer: identification of novel and recurrent deletions by MLPA. Hum Mutat. 2003;22:428–433. - PubMed
-
- Grabowski M, Mueller-Koch Y, Grasbon-Frodl E, Koehler U, Keller G, Vogelsang H, Dietmaier W, Kopp R, Siebers U, Schmitt W, Neitzel B, Gruber M, et al. Deletions account for 17% of pathogenic germline alterations in MLH1 and MSH2 in hereditary nonpolyposis colorectal cancer (HNPCC) families. Genet Test. 2005;9:138–146. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

