Accumulation of oxidative DNA damage in brain mitochondria in mouse model of hereditary ferritinopathy
- PMID: 20478358
- PMCID: PMC2914088
- DOI: 10.1016/j.neulet.2010.05.025
Accumulation of oxidative DNA damage in brain mitochondria in mouse model of hereditary ferritinopathy
Abstract
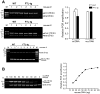
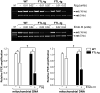
Tissue iron content is strictly regulated to concomitantly satisfy specialized metabolic requirements and avoid toxicity. Ferritin, a multi-subunit iron storage protein, is central to maintenance of iron homeostasis in the brain. Mutations in the ferritin light chain (FTL)-encoding gene underlie the autosomal dominant, neurodegenerative disease, neuroferritinopathy/hereditary ferritinopathy (HF). HF is characterized by progressive accumulation of ferritin and iron. To gain insight into mechanisms by which FTL mutations promote neurodegeneration, a transgenic mouse, expressing human mutant form of FTL, was recently generated. The FTL mouse exhibits buildup of iron in the brain and presents manifestations of oxidative stress reminiscent of the human disease. Here, we asked whether oxidative DNA damage accumulates in the FTL mouse brain. Long-range PCR (L-PCR) amplification-mediated DNA damage detection assays revealed that the integrity of mitochondrial DNA (mtDNA) in the brain was significantly compromised in the 12- but not 6-month-old FTL mice. Furthermore, L-PCR employed in conjunction with DNA modifying enzymes, which target specific DNA adducts, revealed the types of oxidative adducts accumulating in mtDNA in the FTL brain. Consistently with DNA damage predicted to form under conditions of excessive oxidative stress, detected adducts include, oxidized guanines, abasic sites and strand breaks. Elevated mtDNA damage may impair mitochondrial function and brain energetics and in the long term contribute to neuronal loss and exacerbate neurodegeneration in HF.
Copyright 2010 Elsevier Ireland Ltd. All rights reserved.
Figures

Similar articles
-
Neuropathological and biochemical investigation of Hereditary Ferritinopathy cases with ferritin light chain mutation: Prominent protein aggregation in the absence of major mitochondrial or oxidative stress.Neuropathol Appl Neurobiol. 2021 Feb;47(1):26-42. doi: 10.1111/nan.12634. Epub 2020 Jun 19. Neuropathol Appl Neurobiol. 2021. PMID: 32464705
-
A novel neuroferritinopathy mouse model (FTL 498InsTC) shows progressive brain iron dysregulation, morphological signs of early neurodegeneration and motor coordination deficits.Neurobiol Dis. 2015 Sep;81:119-33. doi: 10.1016/j.nbd.2014.10.023. Epub 2014 Nov 4. Neurobiol Dis. 2015. PMID: 25447222 Free PMC article.
-
Abnormal iron metabolism and oxidative stress in mice expressing a mutant form of the ferritin light polypeptide gene.J Neurochem. 2009 May;109(4):1067-78. doi: 10.1111/j.1471-4159.2009.06028.x. Epub 2009 Mar 30. J Neurochem. 2009. PMID: 19519778 Free PMC article.
-
Iron, Ferritin, Hereditary Ferritinopathy, and Neurodegeneration.Front Neurosci. 2019 Dec 11;13:1195. doi: 10.3389/fnins.2019.01195. eCollection 2019. Front Neurosci. 2019. PMID: 31920471 Free PMC article. Review.
-
Cytosolic and mitochondrial ferritins in the regulation of cellular iron homeostasis and oxidative damage.Biochim Biophys Acta. 2010 Aug;1800(8):783-92. doi: 10.1016/j.bbagen.2010.02.005. Epub 2010 Feb 20. Biochim Biophys Acta. 2010. PMID: 20176086 Review.
Cited by
-
Effect of Systemic Iron Overload and a Chelation Therapy in a Mouse Model of the Neurodegenerative Disease Hereditary Ferritinopathy.PLoS One. 2016 Aug 30;11(8):e0161341. doi: 10.1371/journal.pone.0161341. eCollection 2016. PLoS One. 2016. PMID: 27574973 Free PMC article.
-
Abnormal iron metabolism in fibroblasts from a patient with the neurodegenerative disease hereditary ferritinopathy.Mol Neurodegener. 2010 Nov 10;5:50. doi: 10.1186/1750-1326-5-50. Mol Neurodegener. 2010. PMID: 21067605 Free PMC article.
-
Neurodegeneration with Brain Iron Accumulation Disorders: Valuable Models Aimed at Understanding the Pathogenesis of Iron Deposition.Pharmaceuticals (Basel). 2019 Feb 9;12(1):27. doi: 10.3390/ph12010027. Pharmaceuticals (Basel). 2019. PMID: 30744104 Free PMC article. Review.
-
Pathogenic implications of iron accumulation in multiple sclerosis.J Neurochem. 2012 Jan;120(1):7-25. doi: 10.1111/j.1471-4159.2011.07536.x. Epub 2011 Nov 11. J Neurochem. 2012. PMID: 22004421 Free PMC article. Review.
-
Excess iron harms the brain: the syndromes of neurodegeneration with brain iron accumulation (NBIA).J Neural Transm (Vienna). 2013 Apr;120(4):695-703. doi: 10.1007/s00702-012-0922-8. Epub 2012 Dec 2. J Neural Transm (Vienna). 2013. PMID: 23212724 Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

