The oligosaccharide of gonococcal lipooligosaccharide contains several epitopes that are recognized by human antibodies
- PMID: 20479085
- PMCID: PMC2897362
- DOI: 10.1128/IAI.01445-09
The oligosaccharide of gonococcal lipooligosaccharide contains several epitopes that are recognized by human antibodies
Abstract
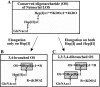
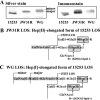
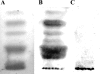
Recently, we isolated human IgG from normal human sera (NHS) using lipooligosaccharide (LOS) from gonococcal strain JW31R as an affinity ligand. We provided evidence that the oligosaccharide (OS) moiety of LOS was immunogenic in humans and that NHS contains functional antibodies that bind to the branched OS. The present study aimed to identify bactericidal antibodies that bind to partial core OS structures or their adjacent sites expressed in the 3,4-branched and 2,3:3,4-dibranched neisserial LOSs. Using 15253 LOS from serum-resistant gonococcal strain 15253 as an affinity ligand, we isolated IgG2 and found that this preparation contained at least three different species. (i) One IgG2 species recognized a cross-reactive epitope that is expressed on 3,4-branched and 2,3:3,4-dibranched neisserial LOSs. (ii) Another IgG2 species was specific for JW31R LOS from a pyocin-resistant gonococcal strain; this IgG-defined epitope was not shared with the aforementioned branched LOSs. (iii) The third IgG2 species bound to the "Salmonella minnesota" Rb and Re mutant lipopolysaccharides (LPSs); this IgG2 recognizes a KDOalpha2-4KDO residue at the reducing end of the carbohydrate moiety of each LPS. The IgG2 was also found to be functional and facilitated the killing of strain 15253. The current results show that neisserial LOS contains several epitopes within its OS moiety that are recognized by human antibodies.
Figures


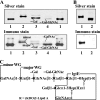

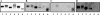
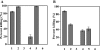
Similar articles
-
Neisseria gonorrhoeae lipooligosaccharide glycan epitopes recognized by bactericidal IgG antibodies elicited by the meningococcal group B-directed vaccine, MenB-4C.Front Immunol. 2024 Feb 19;15:1350344. doi: 10.3389/fimmu.2024.1350344. eCollection 2024. Front Immunol. 2024. PMID: 38440731 Free PMC article.
-
Normal human sera contain bactericidal IgG that binds to the oligosaccharide epitope expressed within lipooligosaccharides of Neisseria gonorrhoeae.J Biochem. 2005 Apr;137(4):487-94. doi: 10.1093/jb/mvi061. J Biochem. 2005. PMID: 15858172
-
Preclinical Efficacy of a Lipooligosaccharide Peptide Mimic Candidate Gonococcal Vaccine.mBio. 2019 Nov 5;10(6):e02552-19. doi: 10.1128/mBio.02552-19. mBio. 2019. PMID: 31690678 Free PMC article.
-
Targeting Lipooligosaccharide (LOS) for a Gonococcal Vaccine.Front Immunol. 2019 Feb 27;10:321. doi: 10.3389/fimmu.2019.00321. eCollection 2019. Front Immunol. 2019. PMID: 30873172 Free PMC article. Review.
-
Strategies for mimicking Neisserial saccharide epitopes as vaccines.Int Rev Immunol. 2001;20(2):229-50. doi: 10.3109/08830180109043036. Int Rev Immunol. 2001. PMID: 11878767 Review.
Cited by
-
Human Immune Responses and the Natural History of Neisseria gonorrhoeae Infection.Front Immunol. 2019 Feb 19;9:3187. doi: 10.3389/fimmu.2018.03187. eCollection 2018. Front Immunol. 2019. PMID: 30838004 Free PMC article. Review.
-
Functional characterization of the gonococcal polyphosphate pseudo-capsule.PLoS Pathog. 2023 May 22;19(5):e1011400. doi: 10.1371/journal.ppat.1011400. eCollection 2023 May. PLoS Pathog. 2023. PMID: 37216411 Free PMC article.
-
Neisseria gonorrhoeae lipooligosaccharide glycan epitopes recognized by bactericidal IgG antibodies elicited by the meningococcal group B-directed vaccine, MenB-4C.Front Immunol. 2024 Feb 19;15:1350344. doi: 10.3389/fimmu.2024.1350344. eCollection 2024. Front Immunol. 2024. PMID: 38440731 Free PMC article.
-
Potential targets for next generation antimicrobial glycoconjugate vaccines.FEMS Microbiol Rev. 2018 May 1;42(3):388-423. doi: 10.1093/femsre/fuy011. FEMS Microbiol Rev. 2018. PMID: 29547971 Free PMC article. Review.
References
-
- Estabrook, M. M., C. J. Baker, and J. M. Griffiss. 1993. The immune response of children to meningococcal lipooligosaccharides during disseminated disease is directed primarily against two monoclonal antibody-defined epitopes. J. Infect. Dis. 167:966-970. - PubMed
-
- Gamian, A., M. Beurret, F. Michon, J. R. Brisson, and H. J. Jennings. 1992. Structure of the L2 lipopolysaccharide core oligosaccharides of Neisseria meningitidis. J. Biol. Chem. 267:922-925. - PubMed
-
- Gibson, B. W., J. W. Webb, R. Yamasaki, S. J. Fisher, A. L. Burlingame, R. E. Mandrell, H. Schneider, and J. M. Griffiss. 1989. Structure and heterogeneity of the oligosaccharides from the lipopolysaccharides of a pyocin-resistant Neisseria gonorrhoeae. Proc. Natl. Acad. Sci. U. S. A. 86:17-21. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

