Serum anti-BPAG1 auto-antibody is a novel marker for human melanoma
- PMID: 20479946
- PMCID: PMC2866734
- DOI: 10.1371/journal.pone.0010566
Serum anti-BPAG1 auto-antibody is a novel marker for human melanoma
Abstract
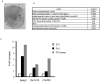
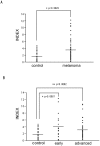
Malignant melanoma is one of the most aggressive types of tumor. Because malignant melanoma is difficult to treat once it has metastasized, early detection and treatment are essential. The search for reliable biomarkers of early-stage melanoma, therefore, has received much attention. By using a novel method of screening tumor antigens and their auto-antibodies, we identified bullous pemphigoid antigen 1 (BPAG1) as a melanoma antigen recognized by its auto-antibody. BPAG1 is an auto-antigen in the skin disease bullous pemphigoid (BP) and anti-BPAG1 auto-antibodies are detectable in sera from BP patients and are used for BP diagnosis. However, BPAG1 has been viewed as predominantly a keratinocyte-associated protein and a relationship between BPAG1 expression and melanoma has not been previously reported. In the present study, we show that bpag1 is expressed in the mouse F10 melanoma cell line in vitro and F10 melanoma tumors in vivo and that BPAG1 is expressed in human melanoma cell lines (A375 and G361) and normal human melanocytes. Moreover, the levels of anti-BPAG1 auto-antibodies in the sera of melanoma patients were significantly higher than in the sera of healthy volunteers (p<0.01). Furthermore, anti-BPAG1 auto-antibodies were detected in melanoma patients at both early and advanced stages of disease. Here, we report anti-BPAG1 auto-antibodies as a promising marker for the diagnosis of melanoma, and we discuss the significance of the detection of such auto-antibodies in cancer biology and patients.
Conflict of interest statement
Figures

References
-
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71–96. - PubMed
-
- Bosserhoff AK. Novel biomarkers in malignant melanoma. Clin Chim Acta. 2006;367:28–35. - PubMed
-
- Ohsie SJ, Sarantopoulos GP, Cochran AJ, Binder SW. Immunohistochemical characteristics of melanoma. J Cutan Pathol. 2008;35:433–444. - PubMed
-
- Brochez L, Naeyaert JM. Serological markers for melanoma. Br J Dermatol. 2000;143:256–268. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

