The halogenated metabolism of brown algae (Phaeophyta), its biological importance and its environmental significance
- PMID: 20479964
- PMCID: PMC2866472
- DOI: 10.3390/md8040988
The halogenated metabolism of brown algae (Phaeophyta), its biological importance and its environmental significance
Abstract
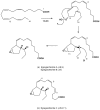
Brown algae represent a major component of littoral and sublittoral zones in temperate and subtropical ecosystems. An essential adaptive feature of this independent eukaryotic lineage is the ability to couple oxidative reactions resulting from exposure to sunlight and air with the halogenations of various substrates, thereby addressing various biotic and abiotic stresses i.e., defense against predators, tissue repair, holdfast adhesion, and protection against reactive species generated by oxidative processes. Whereas marine organisms mainly make use of bromine to increase the biological activity of secondary metabolites, some orders of brown algae such as Laminariales have also developed a striking capability to accumulate and to use iodine in physiological adaptations to stress. We review selected aspects of the halogenated metabolism of macrophytic brown algae in the light of the most recent results, which point toward novel functions for iodide accumulation in kelps and the importance of bromination in cell wall modifications and adhesion properties of brown algal propagules. The importance of halogen speciation processes ranges from microbiology to biogeochemistry, through enzymology, cellular biology and ecotoxicology.
Keywords: biogeochemistry; brown algae; defense metabolites; halogen speciation; haloperoxidases.
Figures
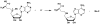

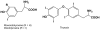
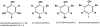




References
-
- Gribble G. The diversity of naturally produced organohalogens. Chemosphere. 2003;52:289–297. - PubMed
-
- Bernroitner M, Zamocky M, Furtmüller PG, Peschek GA, Obinger C. Occurrence, phylogeny, structure, and function of catalases and peroxidases in cyanobacteria. J Exp Bot. 2009;60:423–440. - PubMed
-
- Palmer JD. The symbiotic birth and spread of plastids: how many times and whodunit. J Phycol. 2003;39:4–11.
-
- Phillips N, Calhoun S, Moustafa A, Bhattacharia D, Braun EL. Genomic insights into evolutionary relationships among heterokont lineages emphasizing the Phaeophyceae. J Phycol. 2008;44:15–18. - PubMed
-
- Harper MK, Bugni TS, Copp BR, James RD, Lindsay BS, Richardson AD, Schnabel PC, Tasdemir D, VanWagoner RM, Verbitzki SM, Ireland CM. Introduction to the chemical ecology of marine natural products. In: McClintock JB, Baker BJ, editors. Marine Chemical Ecology. CRC; Boca Raton, FL, USA: 2001. pp. 3–71.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
