Microtubule-stabilizing drugs from marine sponges: focus on peloruside A and zampanolide
- PMID: 20479967
- PMCID: PMC2866475
- DOI: 10.3390/md8041059
Microtubule-stabilizing drugs from marine sponges: focus on peloruside A and zampanolide
Abstract
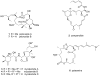
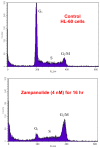
Marine sponges are an excellent source of bioactive secondary metabolites with potential therapeutic value in the treatment of diseases. One group of compounds of particular interest is the microtubule-stabilizing agents, the most well-known compound of this group being paclitaxel (Taxol), an anti-cancer compound isolated from the bark and leaves of the Pacific yew tree. This review focuses on two of the more recent additions to this important class of drugs, peloruside A and zampanolide, both isolated from marine sponges. Peloruside A was isolated from Mycale hentscheli collected in New Zealand coastal waters, and it already shows promising anti-cancer activity. Two other potent bioactive compounds with different modes of action but isolated from the same sponge, mycalamide A and pateamine, will also be discussed. The fourth compound, zampanolide, most recently isolated from the Tongan sponge Cacospongia mycofijiensis, has only recently been added to the microtubule-stabilizing group of compounds, and further work is in progress to determine its activity profile relative to peloruside A and other drugs of this class.
Keywords: microtubule stabilization; mycalamide; pateamine; peloruside; zampanolide.
Figures



Similar articles
-
Peloruside B, a potent antitumor macrolide from the New Zealand marine sponge Mycale hentscheli: isolation, structure, total synthesis, and bioactivity.J Org Chem. 2010 Jan 1;75(1):2-10. doi: 10.1021/jo9021265. J Org Chem. 2010. PMID: 19957922 Free PMC article.
-
Peloruside A: a lead non-taxoid-site microtubule-stabilizing agent with potential activity against cancer, neurodegeneration, and autoimmune disease.Nat Prod Rep. 2016 Apr;33(4):549-61. doi: 10.1039/c5np00146c. Epub 2016 Feb 12. Nat Prod Rep. 2016. PMID: 26867978 Review.
-
Peloruside A: a potent cytotoxic macrolide isolated from the new zealand marine sponge Mycale sp.J Org Chem. 2000 Jan 28;65(2):445-9. doi: 10.1021/jo991296y. J Org Chem. 2000. PMID: 10813954
-
Peloruside A, a novel antimitotic agent with paclitaxel-like microtubule- stabilizing activity.Cancer Res. 2002 Jun 15;62(12):3356-60. Cancer Res. 2002. PMID: 12067973
-
Zampanolide and dactylolide: cytotoxic tubulin-assembly agents and promising anticancer leads.Nat Prod Rep. 2014 Sep;31(9):1202-26. doi: 10.1039/c4np00024b. Nat Prod Rep. 2014. PMID: 24945566 Free PMC article. Review.
Cited by
-
Ethyl acetate extract from marine sponge Hyattella cribriformis exhibit potent anticancer activity by promoting tubulin polymerization as evidenced mitotic arrest and induction of apoptosis.Pharmacogn Mag. 2015 Apr-Jun;11(42):345-55. doi: 10.4103/0973-1296.153088. Pharmacogn Mag. 2015. PMID: 25829774 Free PMC article.
-
Drug discovery targeting cell division proteins, microtubules and FtsZ.Bioorg Med Chem. 2014 Sep 15;22(18):5060-77. doi: 10.1016/j.bmc.2014.02.036. Epub 2014 Mar 5. Bioorg Med Chem. 2014. PMID: 24680057 Free PMC article. Review.
-
Bioactive substances with anti-neoplastic efficacy from marine invertebrates: Porifera and Coelenterata.World J Clin Oncol. 2011 Nov 10;2(11):355-61. doi: 10.5306/wjco.v2.i11.355. World J Clin Oncol. 2011. PMID: 22087433 Free PMC article.
-
Metabolomic tools for secondary metabolite discovery from marine microbial symbionts.Mar Drugs. 2014 Jun 5;12(6):3416-48. doi: 10.3390/md12063416. Mar Drugs. 2014. PMID: 24905482 Free PMC article.
-
Axonal Transport and Neurodegeneration: How Marine Drugs Can Be Used for the Development of Therapeutics.Mar Drugs. 2016 May 19;14(5):102. doi: 10.3390/md14050102. Mar Drugs. 2016. PMID: 27213408 Free PMC article. Review.
References
-
- Paterson I, Anderson EA. The renaissance of natural products as drug candidates. Science. 2005;310:451–453. - PubMed
-
- Newman D, Cragg GM. Natural products as sources of new drugs over the last 25 years. J Nat Prod. 2007;70:461–477. - PubMed
-
- Molinski TF, Dalisay DS, Lievens SL, Saludes JP. Drug development from marine natural products. Nat Rev Drug Discov. 2009;8:69–85. - PubMed
-
- Cragg GM, Grothaus PG, Newman DJ. Impact of natural products on developing new anti-cancer agents. Chem Rev. 2009;109:3012–3043. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
