Donor-acceptor-donor fluorene derivatives for two-photon fluorescence lysosomal imaging
- PMID: 20481577
- PMCID: PMC2888463
- DOI: 10.1021/jo100554j
Donor-acceptor-donor fluorene derivatives for two-photon fluorescence lysosomal imaging
Abstract
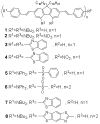
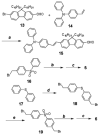
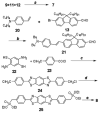
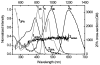
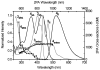
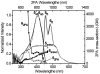
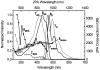
As part of a strategy to achieve large two-photon absorptivity in fluorene-based probes, a series of donor-acceptor-donor (D-A-D) type derivatives were synthesized and their two-photon absorption (2PA) properties investigated. The synthesis of D-A-D fluorophores was achieved by efficient preparation of key intermediates for the introduction of central electron acceptor groups. To accomplish the synthesis of two of the new derivatives, a high-yield method for a one-step direct dibromomethylation of phenyl sulfide was developed. The linear and nonlinear optical properties, including UV-vis absorption, fluorescence emission, fluorescence anisotropy, and two-photon absorption (2PA), of the new D-A-D compounds were measured and compared to their D-A or D-D counterparts. Fully conjugated acceptor moieties in the center of the D-A-D fluorophore led to the greatest increase in the 2PA cross section, while weakly conjugated central acceptors exhibited only a modest increase in the 2PA cross section relative to D-A diploar analogs. Encapsulation of the new probes in Pluronic F 108NF micelles, and subsequent incubation in HCT 116 cells, resulted in very high lysosomal colocalization (>0.98 colocalization coefficient) relative to commercial Lysotracker Red, making the micelle-encapsulated dyes particularly attractive as fluorescent probes for two-photon fluorescence microscopy lysosomal imaging.
Figures
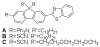



Similar articles
-
A series of fluorene-based two-photon absorbing molecules: synthesis, linear and nonlinear characterization, and bioimaging.J Org Chem. 2010 Jun 18;75(12):3975-82. doi: 10.1021/jo1005075. J Org Chem. 2010. PMID: 20481596 Free PMC article.
-
High-fidelity hydrophilic probe for two-photon fluorescence lysosomal imaging.J Am Chem Soc. 2010 Sep 8;132(35):12237-9. doi: 10.1021/ja1057423. J Am Chem Soc. 2010. PMID: 20712313 Free PMC article.
-
Two-photon STED spectral determination for a new V-shaped organic fluorescent probe with efficient two-photon absorption.Chemphyschem. 2011 Oct 24;12(15):2755-62. doi: 10.1002/cphc.201100456. Epub 2011 Aug 19. Chemphyschem. 2011. PMID: 21858908 Free PMC article.
-
Lighting the Way to See Inside Two-Photon Absorption Materials: Structure-Property Relationship and Biological Imaging.Materials (Basel). 2017 Feb 23;10(3):223. doi: 10.3390/ma10030223. Materials (Basel). 2017. PMID: 28772584 Free PMC article. Review.
-
Roadmap for Designing Donor-π-Acceptor Fluorophores in UV-Vis and NIR Regions: Synthesis, Optical Properties and Applications.Biomolecules. 2025 Jan 14;15(1):119. doi: 10.3390/biom15010119. Biomolecules. 2025. PMID: 39858513 Free PMC article. Review.
Cited by
-
Folate receptor-targeted aggregation-enhanced near-IR emitting silica nanoprobe for one-photon in vivo and two-photon ex vivo fluorescence bioimaging.Bioconjug Chem. 2011 Jul 20;22(7):1438-50. doi: 10.1021/bc2002506. Epub 2011 Jul 1. Bioconjug Chem. 2011. PMID: 21688841 Free PMC article.
-
Tuning the π-bridge of quadrupolar triarylborane chromophores for one- and two-photon excited fluorescence imaging of lysosomes in live cells.Chem Sci. 2019 Apr 23;10(20):5405-5422. doi: 10.1039/c9sc00793h. eCollection 2019 May 28. Chem Sci. 2019. PMID: 31217943 Free PMC article.
-
Tuning the Properties of Donor-Acceptor and Acceptor-Donor-Acceptor Boron Difluoride Hydrazones via Extended π-Conjugation.ACS Omega. 2022 Aug 26;7(36):32727-32739. doi: 10.1021/acsomega.2c04401. eCollection 2022 Sep 13. ACS Omega. 2022. PMID: 36120012 Free PMC article.
-
A denitrogenative palladium-catalyzed cascade for regioselective synthesis of fluorenes.Chem Sci. 2019 Dec 17;11(5):1411-1417. doi: 10.1039/c9sc04062e. Chem Sci. 2019. PMID: 34123265 Free PMC article.
-
Fluorene Based Ferric Complex as Colorimetric and Fluorometric Probe for Highly Selective Detection of CN- and S2- Anions.J Fluoresc. 2021 Sep;31(5):1311-1321. doi: 10.1007/s10895-021-02737-w. Epub 2021 Jun 10. J Fluoresc. 2021. PMID: 34110595
References
-
- Denk W, Strickler JH, Webb WW. Science. 1990;248:73–76. - PubMed
-
- Konig K. J Microsc. 2000;200:83–104. - PubMed
-
- Piston DW. Trends Cell Biol. 1999;9:66–69. - PubMed
-
- Xu C, Williams RM, Zipfel W, Webb WW. Bioimaging. 1996;4:198–207.
-
- Schafer-Hales KJ, Belfield KD, Yao S, Frederiksen PK, Hales JM, Kolattukudy PE. J Biomed Opt. 2005;10:051402-1, 8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

