Studies on Xenopus laevis intestine reveal biological pathways underlying vertebrate gut adaptation from embryo to adult
- PMID: 20482879
- PMCID: PMC2898076
- DOI: 10.1186/gb-2010-11-5-r55
Studies on Xenopus laevis intestine reveal biological pathways underlying vertebrate gut adaptation from embryo to adult
Abstract
Background: To adapt to its changing dietary environment, the digestive tract is extensively remodeled from the embryo to the adult during vertebrate development. Xenopus laevis metamorphosis is an excellent model system for studying mammalian gastrointestinal development and is used to determine the genes and signaling programs essential for intestinal development and maturation.
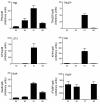
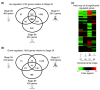
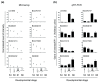
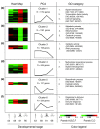
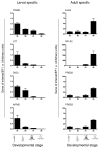
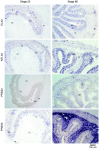
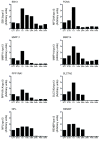
Results: The metamorphosing intestine can be divided into four distinct developmental time points and these were analyzed with X. laevis microarrays. Due to the high level of conservation in developmental signaling programs and homology to mammalian genes, annotations and bioinformatics analysis were based on human orthologs. Clustering of the expression patterns revealed co-expressed genes involved in essential cell processes such as apoptosis and proliferation. The two largest clusters of genes have expression peaks and troughs at the climax of metamorphosis, respectively. Novel conserved gene ontology categories regulated during this period include transcriptional activity, signal transduction, and metabolic processes. Additionally, we identified larval/embryo- and adult-specific genes. Detailed analysis revealed 17 larval specific genes that may represent molecular markers for human colonic cancers, while many adult specific genes are associated with dietary enzymes.
Conclusions: This global developmental expression study provides the first detailed molecular description of intestinal remodeling and maturation during postembryonic development, which should help improve our understanding of intestinal organogenesis and human diseases. This study significantly contributes towards our understanding of the dynamics of molecular regulation during development and tissue renewal, which is important for future basic and clinical research and for medicinal applications.
Figures



References
-
- Dauca M, Bouziges F, Colin S, Kedinger M, Keller MK, Schilt J, Simon-Assmann P, Haffen K. Development of the vertebrate small intestine and mechanisms of cell differentiation. Int J Dev Biol. 1990;34:205–218. - PubMed
-
- Segal GH, Petras RE. In: Histology for Pathologists. Sternberg SS, editor. New York.: Raven Press, Ltd; 1992. Small intestine; pp. 547–571.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

