Mechanisms underlying the antifibrillatory action of hyperkalemia in Guinea pig hearts
- PMID: 20483316
- PMCID: PMC2872203
- DOI: 10.1016/j.bpj.2010.02.011
Mechanisms underlying the antifibrillatory action of hyperkalemia in Guinea pig hearts
Abstract
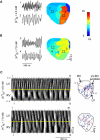
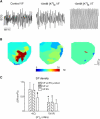
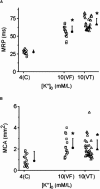
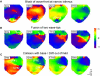
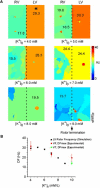
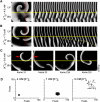
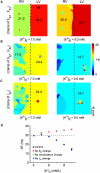
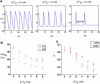
Hyperkalemia increases the organization of ventricular fibrillation (VF) and may also terminate it by mechanisms that remain unclear. We previously showed that the left-to-right heterogeneity of excitation and wave fragmentation present in fibrillating guinea pig hearts is mediated by chamber-specific outward conductance differences in the inward rectifier potassium current (I(K1)). We hypothesized that hyperkalemia-mediated depolarization of the reversal potential of I(K1) (E(K1)) would reduce excitability and thereby reduce VF excitation frequencies and left-to-right heterogeneity. We induced VF in Langendroff-perfused guinea pig hearts and increased the extracellular K(+) concentration ([K(+)](o)) from control (4 mM) to 7 mM (n = 5) or 10 mM (n = 7). Optical mapping enabled spatial characterization of excitation dominant frequencies (DFs) and wavebreaks, and identification of sustained rotors (>4 cycles). During VF, hyperkalemia reduced the maximum DF of the left ventricle (LV) from 31.5 +/- 4.7 Hz (control) to 23.0 +/- 4.7 Hz (7.0 mM) or 19.5 +/- 3.6 Hz (10.0 mM; p < 0.006), the left-to-right DF gradient from 14.7 +/- 3.6 Hz (control) to 4.4 +/- 1.3 Hz (7 mM) and 3.2 +/- 1.4 Hz (10 mM), the number of DF domains, and the incidence of wavebreak in the LV and interventricular regions. During 10 mM [K(+)](o), the rotation period and core area of sustained rotors in the LV increased, and VF often terminated. Two-dimensional computer simulations mimicking experimental VF predicted that clamping E(K1) to normokalemic values during simulated hyperkalemia prevented all of the hyperkalemia-induced VF changes. During hyperkalemia, despite the shortening of the action potential duration, depolarization of E(K1) increased refractoriness, leading to a slowing of VF, which effectively superseded the influence of I(K1) conductance differences on VF organization. This reduced the left-to-right excitation gradients and heterogeneous wavebreak formation. Overall, these results provide, to our knowledge, the first direct mechanistic insight into the organization and/or termination of VF by hyperkalemia.
Copyright 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures
References
-
- Gettes L.S. Electrolyte abnormalities underlying lethal and ventricular arrhythmias. Circulation. 1992;85(1, Suppl):I70–I76. - PubMed
-
- Pitt B., Bakris G., EPHESUS Investigators Serum potassium and clinical outcomes in the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study (EPHESUS) Circulation. 2008;118:1643–1650. - PubMed
-
- Janse M.J., Wit A.L. Electrophysiological mechanisms of ventricular arrhythmias resulting from myocardial ischemia and infarction. Physiol. Rev. 1989;69:1049–1169. - PubMed
-
- Zipes D.P., Wellens H.J. Sudden cardiac death. Circulation. 1998;98:2334–2351. - PubMed
-
- Paterson D.J. Antiarrhythmic mechanisms during exercise. J. Appl. Physiol. 1996;80:1853–1862. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

