α- and β-crystallins modulate the head group order of human lens membranes during aging
- PMID: 20484582
- PMCID: PMC3066618
- DOI: 10.1167/iovs.09-4947
α- and β-crystallins modulate the head group order of human lens membranes during aging
Abstract
Purpose: To examine the physical properties of human lens cell membranes as a function of age.
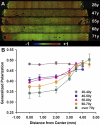
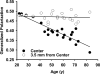
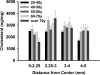
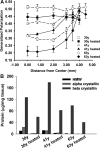
Methods: The environment of the phospholipid head groups in fiber cell membranes from human lenses, aged 22 to 83 years, was assessed with Laurdan and two-photon confocal microscopy. The effect of mild thermal stress on head group order was studied with lens pairs in which one intact lens was incubated at 50 °C. Dihydrosphingomyelin vesicles were preloaded with Laurdan, α-, β-, or γ-crystallin was added, and surface fluidity was determined.
Results: The membrane head group environment became more fluid with age as indicated by increased water penetration. Furthermore, these changes could be replicated simply by exposing intact human lenses to mild thermal stress; conditions which decreased the concentration of soluble α- and β-crystallins. Vesicle binding experiments showed that α- and β-, but not γ-, crystallins markedly affected head group order.
Conclusions: The physical properties of cell membranes in the lens nucleus change substantially with age, and α- and β-crystallins may modulate this effect. β-Crystallins may therefore play a role in lens cells, and cells of other tissues, apart from being simple structural proteins. Age-dependent loss of these crystallins may affect membrane integrity and contribute to the dysfunction of lenses in older people.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

