Advances in molecular imaging with ultrasound
- PMID: 20487678
- PMCID: PMC2935327
Advances in molecular imaging with ultrasound
Abstract
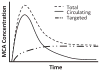
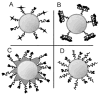
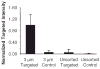
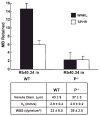
Ultrasound imaging has long demonstrated utility in the study and measurement of anatomic features and noninvasive observation of blood flow. Within the last decade, advances in molecular biology and contrast agents have allowed researchers to use ultrasound to detect changes in the expression of molecular markers on the vascular endothelium and other intravascular targets. This new technology, referred to as ultrasonic molecular imaging, is still in its infancy. However, in preclinical studies, ultrasonic molecular imaging has shown promise in assessing angiogenesis, inflammation, and thrombus. In this review, we discuss recent advances in microbubble-type contrast agent development, ultrasound technology, and signal processing strategies that have the potential to substantially improve the capabilities and utility of ultrasonic molecular imaging.
Figures

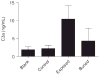

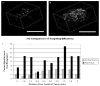
References
-
- Keller MW, Glasheen W, Kaul S. Albunex: a safe and effective commercially produced agent for myocardial contrast echocardiography. J Am Soc Echocardiogr. 1989;2:48–52. - PubMed
-
- Smith MD, Elion JL, McClure RR, et al. Left heart opacification with peripheral venous injection of a new saccharide echo contrast agent in dogs. J Am Coll Cardiol. 1989;13:1622–8. - PubMed
-
- Klibanov AL. Ultrasound contrast agents: development of the field and current status. In: Krause W, editor. Contrast agents II. Vol. 222. New York: Springer-Verlag; 2002. pp. 73–106.
-
- Stride E. Physical principles of microbubbles for ultrasound imaging and therapy. Cerebrovasc Dis. 2009;27 (Suppl 2):1–13. - PubMed
-
- Dayton PA, Ferrara KW. Targeted imaging using ultrasound. J Magn Reson Imaging. 2002;16:362–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
