ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation
- PMID: 20488992
- PMCID: PMC3032591
- DOI: 10.1126/science.1189731
ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation
Abstract
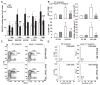
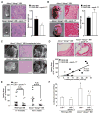
Elevated leukocyte cell numbers (leukocytosis), and monocytes in particular, promote atherosclerosis; however, how they become increased is poorly understood. Mice deficient in the adenosine triphosphate-binding cassette (ABC) transporters ABCA1 and ABCG1, which promote cholesterol efflux from macrophages and suppress atherosclerosis in hypercholesterolemic mice, displayed leukocytosis, a transplantable myeloproliferative disorder, and a dramatic expansion of the stem and progenitor cell population containing Lin(-)Sca-1(+)Kit+ (LSK) in the bone marrow. Transplantation of Abca1(-/-) Abcg1(-/-) bone marrow into apolipoprotein A-1 transgenic mice with elevated levels of high-density lipoprotein (HDL) suppressed the LSK population, reduced leukocytosis, reversed the myeloproliferative disorder, and accelerated atherosclerosis. The findings indicate that ABCA1, ABCG1, and HDL inhibit the proliferation of hematopoietic stem and multipotential progenitor cells and connect expansion of these populations with leukocytosis and accelerated atherosclerosis.
Figures


Comment in
-
Medicine. Tackling two diseases with HDL.Science. 2010 Jun 25;328(5986):1641-2. doi: 10.1126/science.1191663. Science. 2010. PMID: 20576876 No abstract available.
References
-
- Coller BS. Arterioscler Thromb Vasc Biol. 2005;25:658. - PubMed
-
- Gordon DJ, Rifkind BM. N Engl J Med. 1989;321:1311. - PubMed
-
- Barter PJ, Puranik R, Rye KA. Curr Cardiol Rep. 2007;9:493. - PubMed
-
- Tall AR, Yvan-Charvet L, Terasaka N, Pagler T, Wang N. Cell Metab. 2008;7:365. - PubMed
-
- Duffy D, Rader DJ. Nat Rev Cardiol. 2009;6:455. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

