Prostate MRI and 3D MR spectroscopy: how we do it
- PMID: 20489079
- PMCID: PMC2895419
- DOI: 10.2214/AJR.10.4312
Prostate MRI and 3D MR spectroscopy: how we do it
Abstract
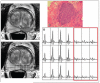
Objective: This review is a primer on the technical aspects of performing a high-quality MRI and MR spectroscopic imaging examination of the prostate.
Conclusion: MRI and MR spectroscopic imaging are useful tools in the localization, staging, and functional assessment of prostate cancer. Performing a high-quality MR spectroscopic examination requires understanding of the technical aspects and limitations of spectral acquisition, postprocessing techniques, and spectral evaluation.
Figures

















References
-
- Heijmink SW, Futterer JJ, Hambrock T, et al. Prostate cancer: body-array versus endorectal coil MR imaging at 3 T—comparison of image quality, localization, and staging performance. Radiology. 2007;244:184–195. - PubMed
-
- Qayyum A, Coakley FV, Lu Y, et al. Organ-confined prostate cancer: effect of prior transrectal biopsy on endorectal MRI and MR spectroscopic imaging. AJR. 2004;183:1079–1083. - PubMed
-
- Claus FG, Hricak H, Hattery RR. Pretreatment evaluation of prostate cancer: role of MR imaging and 1H MR spectroscopy. RadioGraphics. 2004;24(suppl 1):S167–S180. - PubMed
-
- Coakley FV, Kurhanewicz J, Lu Y, et al. Prostate cancer tumor volume: measurement with endorectal MR and MR spectroscopic imaging. Radiology. 2002;223:91–97. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

