Elevated cyclic stretch induces aortic valve calcification in a bone morphogenic protein-dependent manner
- PMID: 20489151
- PMCID: PMC2893650
- DOI: 10.2353/ajpath.2010.090631
Elevated cyclic stretch induces aortic valve calcification in a bone morphogenic protein-dependent manner
Abstract
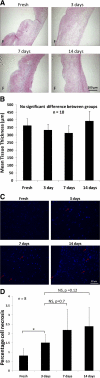
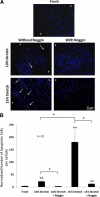
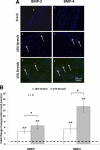
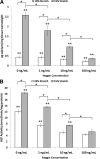
Calcified aortic valve (AV) cusps have increased expression of bone morphogenic proteins (BMPs) and transforming growth factor-beta1 (TGF-beta1). Elevated stretch loading on the AV is known to increase expression of matrix remodeling enzymes and pro-inflammatory proteins. Little, however, is known about the mechanism by which elevated stretch might induce AV calcification. We investigated the hypothesis that elevated stretch may cause valve calcification via a BMP-dependent mechanism. Porcine AV cusps were cultured in a stretch bioreactor, at 10% (physiological) or 15% (pathological) stretch and 70 beats per minute for 3, 7, and 14 days, in osteogenic media supplemented with or without high phosphate (3.8 mmol/L), TGF-beta1 (1 ng/ml), as well as the BMP inhibitor noggin (1, 10, and 100 ng/ml). Fresh cusps served as controls. Alizarin red and von Kossa staining demonstrated that 15% stretch elicited a stronger calcification response compared with 10% stretch in a fully osteogenic medium containing high phosphate and TGF-beta1. BMP-2, -4, and Runx2 expression was observed after 3 days on the fibrosa surface of the valve cusp and was stretch magnitude-dependent. Cellular apoptosis was highest at 15% stretch. Tissue calcium content and alkaline phosphatase activity were similarly stretch-dependent and were significantly reduced by noggin in a dose dependent manner. These results underline the potential role of BMPs in valve calcification due to altered stretch.
Figures





References
-
- Mohler ER., 3rd Mechanisms of aortic valve calcification. Am J Cardiol. 2004;94:1396–1402, A1396. - PubMed
-
- Otto CM, Kuusisto J, Reichenbach DD, Gown AM, O'Brien KD. Characterization of the early lesion of “degenerative” valvular aortic stenosis: histological and immunohistochemical studies. Circulation. 1994;90:844–853. - PubMed
-
- Clark-Greuel JN, Connolly JM, Sorichillo E, Narula NR, Rapoport HS, Mohler ER, 3rd, Gorman JH, 3rd, Gorman RC, Levy RJ. Transforming growth factor-beta1 mechanisms in aortic valve calcification: increased alkaline phosphatase and related events. Ann Thorac Surg. 2007;83:946–953. - PubMed
-
- Jian B, Narula N, Li QY, Mohler ER, 3rd, Levy RJ. Progression of aortic valve stenosis: TGF-beta1 is present in calcified aortic valve cusps and promotes aortic valve interstitial cell calcification via apoptosis. Ann Thorac Surg. 2003;75:457–465. discussion 465–466. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

