Telomeres and telomerase in normal and cancer stem cells
- PMID: 20493857
- PMCID: PMC3370416
- DOI: 10.1016/j.febslet.2010.05.026
Telomeres and telomerase in normal and cancer stem cells
Abstract
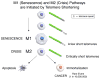
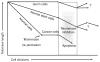
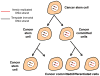
Differences between normal adult tissue stem cells and cancer stem/initiating cells remain poorly defined. For example, it is controversial if cancer stem cells can become fully quiescent, require a stem cell niche, are better at repairing DNA damage than the bulk of the cancer cells, and if and how they regulate symmetric versus asymmetric cell divisions. This minireview will not only provide our personal views to address some of these outstanding questions, but also present evidence that an understanding of telomere dynamics and telomerase activity in normal and cancer stem cells may provide additional insights into how tumors are initiated, and how they should be monitored and treated.
Copyright 2010 Federation of European Biochemical Societies. Published by Elsevier B.V. All rights reserved.
Figures
Similar articles
-
Telomere and telomerase in stem cells: relevance in ageing and disease.Front Biosci (Schol Ed). 2012 Jan 1;4(1):16-30. doi: 10.2741/248. Front Biosci (Schol Ed). 2012. PMID: 22202040 Review.
-
Extension of life-span by introduction of telomerase into normal human cells.Science. 1998 Jan 16;279(5349):349-52. doi: 10.1126/science.279.5349.349. Science. 1998. PMID: 9454332
-
The role of telomeres and telomerase in stem cell aging.FEBS Lett. 2010 Sep 10;584(17):3826-30. doi: 10.1016/j.febslet.2010.07.042. Epub 2010 Aug 3. FEBS Lett. 2010. PMID: 20674573 Review.
-
Changes in telomerase activity and telomere length during human T lymphocyte senescence.Exp Cell Res. 1997 Mar 15;231(2):346-53. doi: 10.1006/excr.1997.3475. Exp Cell Res. 1997. PMID: 9087176
-
Telomeres and telomerase in stem cells during aging and disease.Genome Dyn. 2006;1:84-103. doi: 10.1159/000092502. Genome Dyn. 2006. PMID: 18724055 Review.
Cited by
-
Therapeutic targeting of replicative immortality.Semin Cancer Biol. 2015 Dec;35 Suppl(Suppl):S104-S128. doi: 10.1016/j.semcancer.2015.03.007. Epub 2015 Apr 11. Semin Cancer Biol. 2015. PMID: 25869441 Free PMC article. Review.
-
Enrichment of human osteosarcoma stem cells based on hTERT transcriptional activity.Oncotarget. 2013 Dec;4(12):2326-38. doi: 10.18632/oncotarget.1554. Oncotarget. 2013. PMID: 24334332 Free PMC article.
-
Healthy aging and disease: role for telomere biology?Clin Sci (Lond). 2011 May;120(10):427-40. doi: 10.1042/CS20100385. Clin Sci (Lond). 2011. PMID: 21271986 Free PMC article. Review.
-
Strategies for the Isolation and Identification of Gastric Cancer Stem Cells.Stem Cells Int. 2024 Jun 6;2024:5553852. doi: 10.1155/2024/5553852. eCollection 2024. Stem Cells Int. 2024. PMID: 38882596 Free PMC article. Review.
-
A telomere-targeting drug depletes cancer initiating cells and promotes anti-tumor immunity in small cell lung cancer.Nat Commun. 2024 Jan 22;15(1):672. doi: 10.1038/s41467-024-44861-8. Nat Commun. 2024. PMID: 38253555 Free PMC article.
References
-
- Watson J. Origin of concatemeric T7 DNA. Nat New Biol. 1972;239:197–200. - PubMed
-
- Shay JW, Wright WE. Historical claims and current interpretations of replicative aging. Nat Biotechnol. 2002;20:682–688. - PubMed
-
- Wright WE, Piatyszek MA, Rainey WE, Byrd W, Shay JW. Telomerase activity in human germline and embryonic tissues and cells. Dev Genet. 1995;18:173–179. - PubMed
-
- Shay JW, Wright WE, Werbin H. Defining the molecular mechanism of human cell immortalization. Biochim Biophys Acta. 1991;1072:1–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

