Uncoupling protein-3 lowers reactive oxygen species production in isolated mitochondria
- PMID: 20493945
- PMCID: PMC2903626
- DOI: 10.1016/j.freeradbiomed.2010.05.010
Uncoupling protein-3 lowers reactive oxygen species production in isolated mitochondria
Abstract
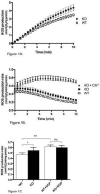
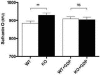
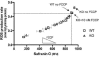
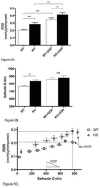
Mitochondria are the major cellular producers of reactive oxygen species (ROS), and mitochondrial ROS production increases steeply with increased proton-motive force. The uncoupling proteins (UCP1, UCP2, and UCP3) and adenine nucleotide translocase induce proton leak in response to exogenously added fatty acids, superoxide, or lipid peroxidation products. "Mild uncoupling" by these proteins may provide a negative feedback loop to decrease proton-motive force and attenuate ROS production. Using wild-type and Ucp3(-/-) mice, we found that native UCP3 actively lowers the rate of ROS production in isolated energized skeletal muscle mitochondria, in the absence of exogenous activators. The estimated specific activity of UCP3 in lowering ROS production was 90 to 500 times higher than that of the adenine nucleotide translocase. The mild uncoupling hypothesis was tested by measuring whether the effect of UCP3 on ROS production could be mimicked by chemical uncoupling. A chemical uncoupler mimicked the effect of UCP3 at early time points after mitochondrial energization, in support of the mild uncoupling hypothesis. However, at later time points the uncoupler did not mimic UCP3, suggesting that UCP3 can also affect ROS production through a membrane potential-independent mechanism.
Copyright (c) 2010 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Cold tolerance of UCP1-ablated mice: a skeletal muscle mitochondria switch toward lipid oxidation with marked UCP3 up-regulation not associated with increased basal, fatty acid- or ROS-induced uncoupling or enhanced GDP effects.Biochim Biophys Acta. 2010 Jun-Jul;1797(6-7):968-80. doi: 10.1016/j.bbabio.2010.02.033. Epub 2010 Mar 19. Biochim Biophys Acta. 2010. PMID: 20227385
-
Stimulation of mitochondrial proton conductance by hydroxynonenal requires a high membrane potential.Biosci Rep. 2008 Apr;28(2):83-8. doi: 10.1042/BSR20080002. Biosci Rep. 2008. PMID: 18384278 Free PMC article.
-
Effect of aging, caloric restriction, and uncoupling protein 3 (UCP3) on mitochondrial proton leak in mice.Exp Gerontol. 2008 Dec;43(12):1069-76. doi: 10.1016/j.exger.2008.09.010. Epub 2008 Sep 30. Exp Gerontol. 2008. PMID: 18852040 Free PMC article.
-
Novel uncoupling proteins.Novartis Found Symp. 2007;287:70-80; discussion 80-91. Novartis Found Symp. 2007. PMID: 18074632 Review.
-
The energetic implications of uncoupling protein-3 in skeletal muscle.Appl Physiol Nutr Metab. 2007 Oct;32(5):884-94. doi: 10.1139/H07-063. Appl Physiol Nutr Metab. 2007. PMID: 18059613 Review.
Cited by
-
Nuclear factor erythroid 2-related factor 2 activation in streptozotocin-induced diabetic rats normalize renal hemodynamics and oxygen consumption.Ups J Med Sci. 2024 Jul 9;129. doi: 10.48101/ujms.v129.10791. eCollection 2024. Ups J Med Sci. 2024. PMID: 39114321 Free PMC article.
-
Production of reactive oxygen species by multipotent stromal cells/mesenchymal stem cells upon exposure to fas ligand.Cell Transplant. 2012;21(10):2171-87. doi: 10.3727/096368912X639035. Epub 2012 Apr 17. Cell Transplant. 2012. PMID: 22526333 Free PMC article.
-
Loss of Intralipid®- but not sevoflurane-mediated cardioprotection in early type-2 diabetic hearts of fructose-fed rats: importance of ROS signaling.PLoS One. 2014 Aug 15;9(8):e104971. doi: 10.1371/journal.pone.0104971. eCollection 2014. PLoS One. 2014. PMID: 25127027 Free PMC article.
-
Relationship of electrophilic stress to aging.Free Radic Biol Med. 2011 Sep 15;51(6):1087-105. doi: 10.1016/j.freeradbiomed.2011.05.039. Epub 2011 Jun 12. Free Radic Biol Med. 2011. PMID: 21708248 Free PMC article. Review.
-
Mediators of Physical Activity Protection against ROS-Linked Skeletal Muscle Damage.Int J Mol Sci. 2019 Jun 20;20(12):3024. doi: 10.3390/ijms20123024. Int J Mol Sci. 2019. PMID: 31226872 Free PMC article. Review.
References
-
- Brand MD, Affourtit C, Esteves TC, Green K, Lambert AJ, Miwa S, Pakay JL, Parker N. Mitochondrial superoxide: production, biological effects, and activation of uncoupling proteins. Free Radic. Biol. Med. 2004;37:755–767. - PubMed
-
- Raha S, Robinson BH. Mitochondria, oxygen free radicals, disease and ageing. Trends Biochem. Sci. 2000;25:502–508. - PubMed
-
- Ambrosio G, Zweier JL, Duilio C, Kuppusamy P, Santoro G, Elia PP, Tritto I, Cirillo P, Condorelli M, Chiariello M, et al. Evidence that mitochondrial respiration is a source of potentially toxic oxygen free radicals in intact rabbit hearts subjected to ischemia and reflow. J. Biol. Chem. 1993;268:18532–18541. - PubMed
-
- St-Pierre J, Buckingham JA, Roebuck SJ, Brand MD. Topology of superoxide production from different sites in the mitochondrial electron transport chain. J. Biol. Chem. 2002;277:44784–44790. - PubMed
-
- Korshunov SS, Skulachev VP, Starkov AA. High protonic potential actuates a mechanism of production of reactive oxygen species in mitochondria. FEBS Lett. 1997;416:15–18. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

