EphB3 limits the expansion of neural progenitor cells in the subventricular zone by regulating p53 during homeostasis and following traumatic brain injury
- PMID: 20496368
- PMCID: PMC2967180
- DOI: 10.1002/stem.449
EphB3 limits the expansion of neural progenitor cells in the subventricular zone by regulating p53 during homeostasis and following traumatic brain injury
Abstract
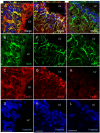
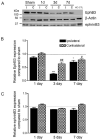
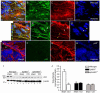
Ephrins and Eph receptor(s) have recently been implicated in regulating neurogenesis in the adult subventricular zone (SVZ) and rostral migratory stream. Here, we examined the role of ephrinB3-EphB3 signaling in mediating the SVZ response to traumatic brain injury (TBI). Analysis of EphB3 expression showed colocalization with glial fibrillary acidic protein-positive neural stem progenitor cells (NSPCs) and doublecortin-positive neuroblasts, whereas ephrinB3 was expressed outside the neurogenic region. TBI resulted in a significant reduction in EphB3 expression, which coincided with enhanced NSPC survival and proliferation at 3 and 7 days postinjury. Analysis of mice lacking either ephrinB3 (ephrinB3(-/-)) or EphB3 (EphB3(-/-)) showed a significant increase in bromodeoxyuridine (BrdU) incorporation and Ki67 immunoreactivity in the SVZ. Interestingly, cell death was dissimilar between knockout mice, where cell death was reduced in EphB3(-/-) but increased in ephrinB3(-/-) mice. Lateral ventricle infusion of soluble preclustered ephrinB3-Fc reversed the proliferative and cell death defects in ephrinB3(-/-) but not EphB3(-/-) mice and prevented TBI-induced proliferation in wild-type NSPCs. Coincidently, tumor suppressor p53 expression was increased following EphB3 stimulation and is reduced in the absence of either EphB3 or ephrinB3. Furthermore, pharmacological inhibition and siRNA knockdown of p53-attenuated ephrinB3-Fc-mediated growth suppression while having no effect on cell death in cultured NSPCs. These data demonstrate that EphB3 signaling suppresses NSPC proliferation in a p53-dependent manner, induces cell death in the absence of ligand stimulation and is transiently reduced in the SVZ to initiate the expansion and survival of endogenous adult NSPCs following TBI.
Figures

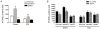

Similar articles
-
Pronounced hypoxia in the subventricular zone following traumatic brain injury and the neural stem/progenitor cell response.Exp Biol Med (Maywood). 2013 Jul;238(7):830-41. doi: 10.1177/1535370213494558. Epub 2013 Jul 4. Exp Biol Med (Maywood). 2013. PMID: 23828590 Free PMC article.
-
EphrinB3 blocks EphB3 dependence receptor functions to prevent cell death following traumatic brain injury.Cell Death Dis. 2014 May 8;5(5):e1207. doi: 10.1038/cddis.2014.165. Cell Death Dis. 2014. PMID: 24810043 Free PMC article.
-
Neural progenitors proliferation is inhibited by EphB3 in the developing subventricular zone.Int J Dev Neurosci. 2011 Feb;29(1):9-14. doi: 10.1016/j.ijdevneu.2010.10.005. Epub 2010 Oct 20. Int J Dev Neurosci. 2011. PMID: 20969945 Free PMC article.
-
Gene profiles within the adult subventricular zone niche: proliferation, differentiation and migration of neural progenitor cells in the ischemic brain.Curr Mol Med. 2007 Aug;7(5):459-62. doi: 10.2174/156652407781387136. Curr Mol Med. 2007. PMID: 17691960 Review.
-
P75 neurotrophin receptor controls subventricular zone neural stem cell migration after stroke.Cell Tissue Res. 2022 Mar;387(3):415-431. doi: 10.1007/s00441-021-03539-z. Epub 2021 Oct 26. Cell Tissue Res. 2022. PMID: 34698916 Free PMC article. Review.
Cited by
-
Modulation of gap junction-associated Cx43 in neural stem/progenitor cells following traumatic brain injury.Brain Res Bull. 2017 Sep;134:38-46. doi: 10.1016/j.brainresbull.2017.06.016. Epub 2017 Jun 23. Brain Res Bull. 2017. PMID: 28648814 Free PMC article.
-
EphrinB3 restricts endogenous neural stem cell migration after traumatic brain injury.Stem Cell Res. 2016 Nov;17(3):504-513. doi: 10.1016/j.scr.2016.09.029. Epub 2016 Sep 28. Stem Cell Res. 2016. PMID: 27771498 Free PMC article.
-
The path from the choroid plexus to the subventricular zone: go with the flow!Front Cell Neurosci. 2012 Aug 9;6:34. doi: 10.3389/fncel.2012.00034. eCollection 2012. Front Cell Neurosci. 2012. PMID: 22907990 Free PMC article.
-
Beyond boundaries--Eph:ephrin signaling in neurogenesis.Cell Adh Migr. 2014;8(4):349-59. doi: 10.4161/19336918.2014.969990. Cell Adh Migr. 2014. PMID: 25482631 Free PMC article. Review.
-
Loss of NLRX1 Exacerbates Neural Tissue Damage and NF-κB Signaling following Brain Injury.J Immunol. 2017 Nov 15;199(10):3547-3558. doi: 10.4049/jimmunol.1700251. Epub 2017 Oct 9. J Immunol. 2017. PMID: 28993512 Free PMC article.
References
-
- Gage FH. Mammalian neural stem cells. Science. 2000;287(5457):1433–8. - PubMed
-
- Kempermann G, Gage FH. New nerve cells for the adult brain. Sci Am. 1999;280(5):48–53. - PubMed
-
- Eriksson PS, et al. Neurogenesis in the adult human hippocampus. Nat Med. 1998;4(11):1313–7. - PubMed
-
- Altman J, Das GD. Autoradiographic and histological studies of postnatal neurogenesis. I. A longitudinal investigation of the kinetics, migration and transformation of cells incorporating tritiated thymidine in neonate rats, with special reference to postnatal neurogenesis in some brain regions. J Comp Neurol. 1966;126(3):337–89. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

