Munc18 and Munc13 regulate early neurite outgrowth
- PMID: 20497124
- PMCID: PMC2895782
- DOI: 10.1042/BC20100036
Munc18 and Munc13 regulate early neurite outgrowth
Abstract
Background information: During development, growth cones of outgrowing neurons express proteins involved in vesicular secretion, such as SNARE (soluble N-ethylmaleimide-sensitive fusion protein-attachment protein receptor) proteins, Munc13 and Munc18. Vesicles are known to fuse in growth cones prior to synapse formation, which may contribute to outgrowth.
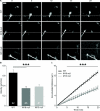
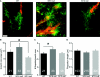
Results: We tested this possibility in dissociated cell cultures and organotypic slice cultures of two release-deficient mice (Munc18-1 null and Munc13-1/2 double null). Both types of release-deficient neurons have a decreased outgrowth speed and therefore have a smaller total neurite length during early development [DIV1-4 (day in vitro 1-4)]. In addition, more filopodia per growth cone were observed in Munc18-1 null, but not WT (wild-type) or Munc13-1/2 double null neurons. The smaller total neurite length during early development was no longer observed after synaptogenesis (DIV14-23).
Conclusion: These data suggest that the inability of vesicle fusion in the growth cone affects outgrowth during the initial phases when outgrowth speed is high, but not during/after synaptogenesis. Overall, the outgrowth speed is probably not rate-limiting during neuronal network formation, at least in vitro. In addition, Munc18, but not Munc13, regulates growth cone filopodia, potentially via its previously observed effect on filamentous actin.
Figures




References
-
- Bostrom P., Andersson L., Rutberg M., Perman J., Lidberg U., Johansson B.R., Fernandez-Rodriguez J., Ericson J., Nilsson T., Boren J., Olofsson S. Snare proteins mediate fusion between cytosolic lipid droplets and are implicated in insulin sensitivity. Nat. Cell Biol. 2007;9:1286–1293. - PubMed
-
- Bouwman J., Maia A.S., Camoletto P.G., Posthuma G., Roubos E.W., Oorschot V.M.J., Klumperman J., Verhage M. Quantification of synapse formation and maintenance in vivo in the absence of synaptic release. Neuroscience. 2004;126:115–126. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

