Permeant anions contribute to voltage dependence of ClC-2 chloride channel by interacting with the protopore gate
- PMID: 20498235
- PMCID: PMC2916987
- DOI: 10.1113/jphysiol.2010.189175
Permeant anions contribute to voltage dependence of ClC-2 chloride channel by interacting with the protopore gate
Abstract
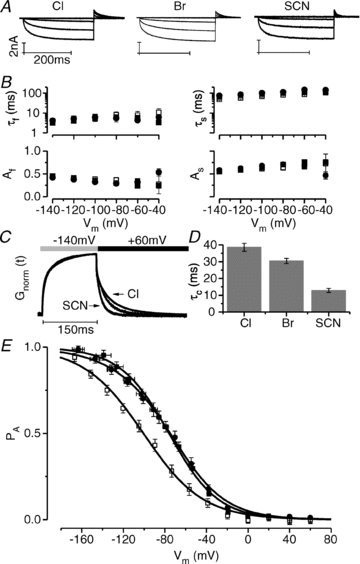
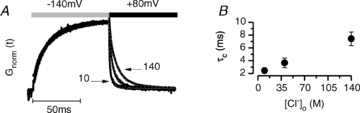
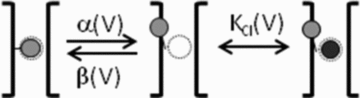
It has been shown that the voltage (V(m)) dependence of ClC Cl(-) channels is conferred by interaction of the protopore gate with H(+) ions. However, in this paper we present evidence which indicates that permeant Cl(-) ions contribute to V(m)-dependent gating of the broadly distributed ClC-2 Cl() channel. The apparent open probability (P(A)) of ClC-2 was enhanced either by changing the [Cl(-)](i) from 10 to 200 mM or by keeping the [Cl(-)](i) low (10 mM) and then raising [Cl(-)](o) from 10 to 140 mM. Additionally, these changes in [Cl(-)] slowed down channel closing at positive V(m) suggesting that high [Cl(-)] increased pore occupancy thus hindering closing of the protopore gate. The identity of the permeant anion was also important since the P(A)(V(m)) curves were nearly identical with Cl(-) or Br(-) but shifted to negative voltages in the presence of SCN() ions. In addition, gating, closing rate and reversal potential displayed anomalous mole fraction behaviour in a SCN(-)/Cl() mixture in agreement with the idea that pore occupancy by different permeant anions modifies the V(m) dependence ClC-2 gating. Based on the ec1-ClC anion pathway, we hypothesized that opening of the protopore gate is facilitated when Cl(-) ions dwell in the central binding site. In contrast, when Cl(-) ions dwell in the external binding site they prevent the gate from closing. Finally, this Cl(-)-dependent gating in ClC-2 channels is of physiological relevance since an increase in [Cl(-)](o) enhances channel opening when the [Cl(-)](i) is in the physiological range.
Figures

 , 40 (▸, ▵), 80 (▿), 140 (•, ▪) and 200 (⋄) m
, 40 (▸, ▵), 80 (▿), 140 (•, ▪) and 200 (⋄) m
References
-
- Bezanilla F. How membrane proteins sense voltage. Nat Rev Mol Cell Biol. 2008;9:323–332. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

