Intervening in global markets to improve access to HIV/AIDS treatment: an analysis of international policies and the dynamics of global antiretroviral medicines markets
- PMID: 20500827
- PMCID: PMC2883977
- DOI: 10.1186/1744-8603-6-9
Intervening in global markets to improve access to HIV/AIDS treatment: an analysis of international policies and the dynamics of global antiretroviral medicines markets
Abstract
Background: Universal access to antiretroviral therapy (ART) in low- and middle-income countries faces numerous challenges: increasing numbers of people needing ART, new guidelines recommending more expensive antiretroviral (ARV) medicines, limited financing, and few fixed-dose combination (FDC) products. Global initiatives aim to promote efficient global ARV markets, yet little is known about market dynamics and the impact of global policy interventions.
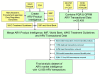
Methods: We utilize several data sources, including 12,958 donor-funded, adult first-line ARV purchase transactions, to describe the market from 2002-2008. We examine relationships between market trends and: World Health Organization (WHO) HIV/AIDS treatment guidelines; WHO Prequalification Programme (WHO Prequal) and United States (US) Food and Drug Administration (FDA) approvals; and procurement policies of the Global Fund to Fight AIDS, Tuberculosis, and Malaria (GFATM), US President's Emergency Plan for AIDS Relief (PEPFAR) and UNITAID.
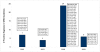
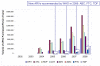
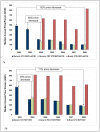
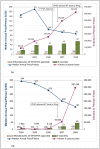
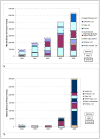
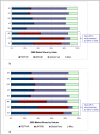
Results: WHO recommended 7, 4, 24, and 6 first-line regimens in 2002, 2003, 2006 and 2009 guidelines, respectively. 2009 guidelines replaced a stavudine-based regimen ($88/person/year) with more expensive zidovudine- ($154-260/person/year) or tenofovir-based ($244-465/person/year) regimens. Purchase volumes for ARVs newly-recommended in 2006 (emtricitabine, tenofovir) increased >15-fold from 2006 to 2008. Twenty-four generic FDCs were quality-approved for older regimens but only four for newer regimens. Generic FDCs were available to GFATM recipients in 2004 but to PEPFAR recipients only after FDA approval in 2006. Price trends for single-component generic medicines mirrored generic FDC prices. Two large-scale purchasers, PEPFAR and UNITAID, together accounted for 53%, 84%, and 77% of market volume for abacavir, emtricitabine, and tenofovir, respectively, in 2008. PEPFAR and UNITAID purchases were often split across two manufacturers.
Conclusions: Global initiatives facilitated the creation of fairly efficient markets for older ARVs, but markets for newer ARVs are less competitive and slower to evolve. WHO guidelines shape demand, and their complexity may help or hinder achievement of economies of scale in pharmaceutical manufacturing. Certification programs assure ARV quality but can delay uptake of new formulations. Large-scale procurement policies may decrease the numbers of buyers and sellers, rendering the market less competitive in the longer-term. Global policies must be developed with consideration for their short- and long-term impact on market dynamics.
Figures

References
-
- World Health Organization, UNAIDS, UNICEF. Towards universal access: Scaling up priority HIV/AIDS interventions in the health sector. Progress report 2009. Geneva: World Health Organization; 2009.
-
- World Health Organization. Rapid advice: antiretroviral therapy for HIV infection in adults and adolescents. Geneva: World Health Organization; 2009.
-
- Paton W. Communication to all CCMs and PRs on 10% savings on round 8. Geneva: Global Fund to Fight AIDS, Tuberculosis and Malaria; 2008.
-
- The Global Fund to Fight AIDS T, and Malaria. Ad Hoc Committees Terms of Reference. Geneva. 2009.
-
- Clinton Foundation. http://www.clintonfoundation.org/
LinkOut - more resources
Full Text Sources

