Identification and functional characterization of the intermediate-conductance Ca(2+)-activated K(+) channel (IK-1) in biliary epithelium
- PMID: 20501432
- PMCID: PMC2777461
- DOI: 10.1152/ajpgi.00223.2009
Identification and functional characterization of the intermediate-conductance Ca(2+)-activated K(+) channel (IK-1) in biliary epithelium
Abstract
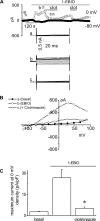
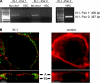
In the liver, adenosine triphosphate (ATP) is an extracellular signaling molecule that is released into bile and stimulates a biliary epithelial cell secretory response via engagement of apical P2 receptors. The molecular identities of the ion channels involved in ATP-mediated secretory responses have not been fully identified. Intermediate-conductance Ca(2+)-activated K(+) channels (IK) have been identified in biliary epithelium, but functional data are lacking. The aim of these studies therefore was to determine the location, function, and regulation of IK channels in biliary epithelial cells and to determine their potential contribution to ATP-stimulated secretion. Expression of IK-1 mRNA was found in both human Mz-Cha-1 biliary cells and polarized normal rat cholangiocyte (NRC) monolayers, and immunostaining revealed membrane localization with a predominant basolateral signal. In single Mz-Cha-1 cells, exposure to ATP activated K(+) currents, increasing current density from 1.6 +/- 0.1 to 7.6 +/- 0.8 pA/pF. Currents were dependent on intracellular Ca(2+) and sensitive to clotrimazole and TRAM-34 (specific IK channel inhibitors). Single-channel recording demonstrated that clotrimazole-sensitive K(+) currents had a unitary conductance of 46.2 +/- 1.5 pS, consistent with IK channels. In separate studies, 1-EBIO (an IK activator) stimulated K(+) currents in single cells that were inhibited by clotrimazole. In polarized NRC monolayers, ATP significantly increased transepithelial secretion which was inhibited by clotrimazole. Lastly, ATP-stimulated K(+) currents were inhibited by the P2Y receptor antagonist suramin and by the inositol 1,4,5-triphosphate (IP3) receptor inhibitor 2-APB. Together these studies demonstrate that IK channels are present in biliary epithelial cells and contribute to ATP-stimulated secretion through a P2Y-IP3 receptor pathway.
Figures







References
-
- Barfod ET, Moore AL, Roe MW, Lidofsky SD. Ca2+-activated IK1 channels associate with lipid rafts upon cell swelling and mediate volume recovery. J Biol Chem 282: 8984–8993, 2007 - PubMed
-
- Basavappa S, Middleton JP, Mangel A, McGill J, Cohn JA, Fitz JG. Cl− and K+ transport in human biliary cell lines. Gastroenterology 104: 1796–1805, 1993 - PubMed
-
- Cohn JA, Strong TA, Picciotto MA, Nairn AC, Collins FS, Francis S, Fitz JG. Localization of CFTR in human bile duct epithelial cells. Gastroenterology 105: 1857–1864, 1993 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

