Attenuation of vaccinia virus by the expression of human Flt3 ligand
- PMID: 20504356
- PMCID: PMC2883979
- DOI: 10.1186/1743-422X-7-109
Attenuation of vaccinia virus by the expression of human Flt3 ligand
Abstract
Background: Vaccinia virus, one of the best known members of poxvirus family, has a wide host range both in vivo and in vitro. The expression of Flt3 ligand (FL) by recombinant vaccinia virus (rVACV) highly influenced properties of the virus in dependence on the level of expression.
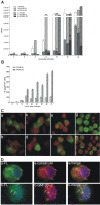
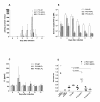
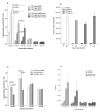
Results: High production of FL driven by the strong synthetic promoter decreased the growth of rVACV in macrophage cell line J774.G8 in vitro as well as its multiplication in vivo when inoculated in mice. The inhibition of replication in vivo was mirrored in low levels of antibodies against vaccinia virus (anti-VACV) which nearly approached to the negative serum level in non-infected mice. Strong FL expression changed not only the host range of the recombinant but also the basic protein contents of virions. The major proteins - H3L and D8L - which are responsible for the virus binding to the cells, and 28 K protein that serves as a virulence factor, were changed in the membrane portion of P13-E/L-FL viral particles. The core virion fraction contained multiple larger, uncleaved proteins and a higher amount of cellular proteins compared to the control virus. The overexpression of FL also resulted in its incorporation into the viral core of P13-E/L-FL IMV particles. In contrary to the equimolar ratio of glycosylated and nonglycosylated FL forms found in cells transfected with the expression plasmid, the recombinant virus incorporated mainly the smaller, nonglycosylated FL.
Conclusions: It has been shown that the overexpression of the Flt3L gene in VACV results in the attenuation of the virus in vivo.
Figures
Similar articles
-
The expression of the soluble isoform of hFlt3 ligand by recombinant vaccinia virus enhances immunogenicity of the vector.Oncol Rep. 2009 May;21(5):1335-43. doi: 10.3892/or_00000359. Oncol Rep. 2009. PMID: 19360312
-
Human Host Range Restriction of the Vaccinia Virus C7/K1 Double Deletion Mutant Is Mediated by an Atypical Mode of Translation Inhibition.J Virol. 2018 Nov 12;92(23):e01329-18. doi: 10.1128/JVI.01329-18. Print 2018 Dec 1. J Virol. 2018. PMID: 30209174 Free PMC article.
-
Primary human macrophages serve as vehicles for vaccinia virus replication and dissemination.J Virol. 2014 Jun;88(12):6819-31. doi: 10.1128/JVI.03726-13. Epub 2014 Apr 2. J Virol. 2014. PMID: 24696488 Free PMC article.
-
Vaccinia virus morphogenesis and dissemination.Trends Microbiol. 2008 Oct;16(10):472-9. doi: 10.1016/j.tim.2008.07.009. Epub 2008 Sep 12. Trends Microbiol. 2008. PMID: 18789694 Review.
-
The interferon system and vaccinia virus evasion mechanisms.J Interferon Cytokine Res. 2009 Sep;29(9):581-98. doi: 10.1089/jir.2009.0073. J Interferon Cytokine Res. 2009. PMID: 19708815 Review.
Cited by
-
Antitumor activity and immunogenicity of recombinant vaccinia virus expressing HPV 16 E7 protein SigE7LAMP is enhanced by high-level coexpression of IGFBP-3.Cancer Gene Ther. 2014 Mar;21(3):115-25. doi: 10.1038/cgt.2014.6. Epub 2014 Feb 21. Cancer Gene Ther. 2014. PMID: 24556712
-
Recombinant Pseudorabies Virus with TK/gE Gene Deletion and Flt3L Co-Expression Enhances the Innate and Adaptive Immune Response via Activating Dendritic Cells.Viruses. 2021 Apr 16;13(4):691. doi: 10.3390/v13040691. Viruses. 2021. PMID: 33923590 Free PMC article.
-
A Recombinant Rabies Virus Expressing Fms-like Tyrosine Kinase 3 Ligand (Flt3L) Induces Enhanced Immunogenicity in Mice.Virol Sin. 2019 Dec;34(6):662-672. doi: 10.1007/s12250-019-00144-x. Epub 2019 Jun 28. Virol Sin. 2019. PMID: 31254272 Free PMC article.
References
-
- McKenna HJ, de Vries P, Brasel K, Lyman SD, Williams DE. Effect of Flt3 Ligand on the Ex Vivo Expansion of Human CD34+ Hematopoietic Progenitor Cells. Blood. 1995;86:3413–3420. - PubMed
-
- Lyman SD, James L, Vanden Bos T, de Vries P, Brasel K, Gliniak B, Hollingsworth LT, Picha KS, McKenna HJ, Splett RR. Molecular Cloning of a Ligand for the Flt3/Flk-2 Tyrosine Kinase Receptor: a Proliferative Factor for Primitive Hematopoietic Cells. Cell. 1993;75:1157–1167. doi: 10.1016/0092-8674(93)90325-K. - DOI - PubMed
-
- Hudak S, Hunte B, Culpepper J, Menon S, Hannum C, Thompson-Snipes L, Rennick D. FLT3/FLK2 Ligand Promotes the Growth of Murine Stem Cells and the Expansion of Colony-Forming Cells and Spleen Colony-Forming Units. Blood. 1995;85:2747–2755. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

