Planetary organic chemistry and the origins of biomolecules
- PMID: 20504964
- PMCID: PMC2890202
- DOI: 10.1101/cshperspect.a003467
Planetary organic chemistry and the origins of biomolecules
Abstract
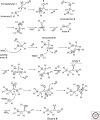
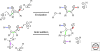
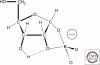
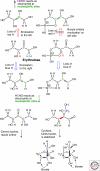
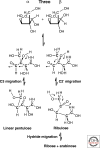
Organic chemistry on a planetary scale is likely to have transformed carbon dioxide and reduced carbon species delivered to an accreting Earth. According to various models for the origin of life on Earth, biological molecules that jump-started Darwinian evolution arose via this planetary chemistry. The grandest of these models assumes that ribonucleic acid (RNA) arose prebiotically, together with components for compartments that held it and a primitive metabolism that nourished it. Unfortunately, it has been challenging to identify possible prebiotic chemistry that might have created RNA. Organic molecules, given energy, have a well-known propensity to form multiple products, sometimes referred to collectively as "tar" or "tholin." These mixtures appear to be unsuited to support Darwinian processes, and certainly have never been observed to spontaneously yield a homochiral genetic polymer. To date, proposed solutions to this challenge either involve too much direct human intervention to satisfy many in the community, or generate molecules that are unreactive "dead ends" under standard conditions of temperature and pressure. Carbohydrates, organic species having carbon, hydrogen, and oxygen atoms in a ratio of 1:2:1 and an aldehyde or ketone group, conspicuously embody this challenge. They are components of RNA and their reactivity can support both interesting spontaneous chemistry as part of a "carbohydrate world," but they also easily form mixtures, polymers and tars. We describe here the latest thoughts on how on this challenge, focusing on how it might be resolved using minerals containing borate, silicate, and molybdate, inter alia.
Figures
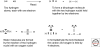

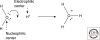
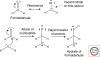
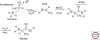





References
-
- Angyal SJ 2001. The Lobry de Bruyn-Alberda van Ekenstein transformation and related reactions. Glycoscience 215:1–14
-
- Benner SA 2009. Life, the Universe and the Scientific Method Gainesville FL, Foundation Press; 320
-
- Bernstein MP, Dworkin J, Sandford SA, Cooper GW, Allamandola LJ 2002. Racemic amino acids from the ultraviolet photolysis of interstellar ice analogues. Nature 416:401–403 - PubMed
-
- Breslow R 1959. On the mechanism of the formose reaction. Tetrahedron Lett 21:22–26
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
