Menopausal symptom experience before and after stopping estrogen therapy in the Women's Health Initiative randomized, placebo-controlled trial
- PMID: 20505547
- PMCID: PMC3770143
- DOI: 10.1097/gme.0b013e3181d76953
Menopausal symptom experience before and after stopping estrogen therapy in the Women's Health Initiative randomized, placebo-controlled trial
Abstract
Objective: The aim of this study was to assess vasomotor and other menopausal symptoms before starting estrogens or placebo, 1 year later, again at trial closure, and after stopping estrogens or placebo. The role of baseline symptoms and age was examined, as was the frequency and determinants of hormone use and symptom management strategies after discontinuing conjugated equine estrogens (CEE) or placebo.
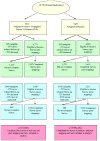
Methods: Intent-to-treat analyses of 10,739 postmenopausal women before and 1 year after randomization to CEE or placebo at 40 clinical centers and a cohort analysis of participants (n = 3,496) who continued taking assigned study pills up to trial closure and completed symptom surveys shortly before (mean, 7.4 +/- 1.1 y from baseline) and after (mean, 306 +/- 55 d after trial closure) stopping pills were performed. Generalized linear regression modeled vasomotor symptoms, vaginal dryness, breast tenderness, pain/stiffness, and mood swings as a function of treatment assignment and baseline symptoms, before and after stopping study pills.
Results: Approximately one third of participants reported at least one moderate to severe symptom at baseline. Fewer symptoms were reported with increasing age, except joint pain/stiffness, which was similar among age groups. At 1 year, hot flashes, night sweats, and vaginal dryness were reduced by CEE, whereas breast tenderness was increased. Breast tenderness was also significantly higher in the CEE group at trial closure. After stopping, vasomotor symptoms were reported by significantly more women who had reported symptoms at baseline, compared with those who had not, and by significantly more participants assigned to CEE (9.8%) versus placebo (3.2%); however, among women with no moderate or severe symptoms at baseline, more than five times as many reported hot flashes after stopping CEE (7.2%) versus placebo (1.5%).
Conclusions: CEE significantly reduced vasomotor symptoms and vaginal dryness in women with baseline symptoms but increased breast tenderness. The likelihood of experiencing symptoms was significantly higher after stopping CEE than placebo regardless of baseline symptom status. These potential effects should be considered before initiating CEE to relieve menopausal symptoms.
Figures
Comment in
-
In search of a middle ground: hormone therapy and its role in modern menopause management.Menopause. 2010 Sep-Oct;17(5):898-9. doi: 10.1097/gme.0b013e3181f1853a. Menopause. 2010. PMID: 20717059 No abstract available.
-
To the Editor. Re: Menopausal symptom experience before and after stopping estrogen therapy in the Women's Health Initiative randomized, placebo-controlled trial.Menopause. 2010 Nov-Dec;17(6):1207; author reply 1207-8. doi: 10.1097/gme.0b013e3181f7dd16. Menopause. 2010. PMID: 21060264 No abstract available.
References
-
- Writing Group for the Women’s Health Initiative. Risks and benefits of estrogen plus progestin in health postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321–333. - PubMed
-
- FDA Updates Menopausal Hormone Information for Postmenopausal Women. US Department of Health and Human Services. US Food and Drug Administration (website); Feb 10, 2004.
-
- Haskell SG. After the Women’s Health Initiative: Postmenopausal women’s experiences with discontinuing estrogen replacement therapy. J Womens Health (Larchmt) 2004;13:438–42. - PubMed
-
- Ness J, Aronow WS, Beck G. Menopausal symptoms after cessation of hormone replacement therapy. Maturitas. 2006;53:356–61. - PubMed
-
- Schonberg MA, Wee CC. Menopausal symptom management and prevention counseling after the Women’s Health Initiative among women seen in an internal medicine practice. J Womens Health (Larchmt) 2005;14:507–14. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

