An inducible caspase 9 suicide gene to improve the safety of mesenchymal stromal cell therapies
- PMID: 20506146
- PMCID: PMC3290395
- DOI: 10.1002/stem.433
An inducible caspase 9 suicide gene to improve the safety of mesenchymal stromal cell therapies
Abstract
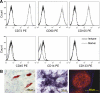
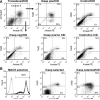
Mesenchymal stromal cells (MSCs) have been infused in hundreds of patients to date, with minimal reported side effects. However, follow-up is limited and long-term side effects are unknown. Because several animal models have raised safety concerns, we sought to develop a system allowing control over the growth and survival of MSCs used therapeutically. We have previously described a suicide system based on an inducible caspase-9 (iCasp9) protein that is activated using a specific chemical inducer of dimerization (CID), analogs of which have been safely tested in a phase I study. Here, we show that MSCs can be easily transduced with this system and selected to high purity (greater than 97%) with clinical grade immunomagnetic procedures. The transduced cells maintain their basic physiology, including expression of surface antigens (such as positivity for CD73, CD90, and CD105, and negativity for hematopoietic markers) and their potential to differentiate into diverse connective tissue lineages (adipocytes, osteoblasts, and chondroblasts). Those cells and their differentiated progeny can be selectively eliminated in vitro or in vivo within 24 hours after exposure to pharmacological levels of CID, with evidence of apoptosis in more than 95% of iCasp9-positive cells. In conclusion, we have developed directed MSC killing to provide a necessary safety mechanism for therapies using progenitor cells. We believe that this approach will become of increasing value as clinical applications for MSCs develop further.
Figures



References
-
- Pittenger MF, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. - PubMed
-
- Dominici M, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315–317. - PubMed
-
- Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276:71–74. - PubMed
-
- Studeny M, Marini FC, Champlin RE, Zompetta C, Fidler IJ, Andreeff M. Bone marrow-derived mesenchymal stem cells as vehicles for interferon-beta delivery into tumors. Cancer Res. 2002;62:3603–3608. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

