Effects of acute cocaine or dopamine receptor agonists on AMPA receptor distribution in the rat nucleus accumbens
- PMID: 20506566
- PMCID: PMC2965794
- DOI: 10.1002/syn.20823
Effects of acute cocaine or dopamine receptor agonists on AMPA receptor distribution in the rat nucleus accumbens
Abstract
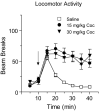
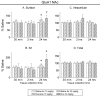
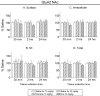
Changes in α-amino-3-hydroxy-5-methylisoxazole-4-propionate receptor (AMPAR) surface expression in the rodent nucleus accumbens (NAc) are produced by cocaine exposure and implicated in addiction-related behaviors. The direction of change depends on the animal's prior drug history. However, little is known about the effect of a single exposure to cocaine on AMPAR distribution in the NAc of untreated rats. This is essential information for interpreting the literature on AMPAR trafficking after repeated cocaine exposure. In this study, we used a protein crosslinking assay to determine the effect of a single cocaine injection on surface and intracellular AMPAR subunit levels in the rat NAc. We found increased AMPAR surface expression in the NAc 24 h, but not 30 min or 2 h, after cocaine injection. A major effect of cocaine is to increase extracellular dopamine (DA) levels, leading to DA receptor activation. Therefore, we also evaluated the effects of directly acting DA receptor agonists. In contrast to the effects of cocaine, AMPAR surface expression was significantly decreased 24 h after injection of the D2-class agonist quinpirole, whereas no significant effects were produced by the D1-class agonist SKF 81297 or the mixed DA agonist apomorphine. Our results show that the effects of a single cocaine exposure in drug- and injection-naïve rats are distinct from those previously reported after repeated cocaine administration. They further suggest that cocaine exerts these effects by influencing neuronal circuits rather than simply stimulating NAc DA transmission.
Copyright © 2010 Wiley-Liss, Inc.
Figures



Similar articles
-
AMPA receptor plasticity in the nucleus accumbens after repeated exposure to cocaine.Neurosci Biobehav Rev. 2010 Nov;35(2):185-211. doi: 10.1016/j.neubiorev.2010.01.013. Epub 2010 Jan 28. Neurosci Biobehav Rev. 2010. PMID: 20109488 Free PMC article. Review.
-
Renewed cocaine exposure produces transient alterations in nucleus accumbens AMPA receptor-mediated behavior.J Neurosci. 2008 Nov 26;28(48):12808-14. doi: 10.1523/JNEUROSCI.2060-08.2008. J Neurosci. 2008. PMID: 19036973 Free PMC article.
-
Acute and chronic dopamine receptor stimulation modulates AMPA receptor trafficking in nucleus accumbens neurons cocultured with prefrontal cortex neurons.J Neurosci. 2008 Apr 16;28(16):4216-30. doi: 10.1523/JNEUROSCI.0258-08.2008. J Neurosci. 2008. PMID: 18417701 Free PMC article.
-
Adenosine A1 and dopamine d1 receptor regulation of AMPA receptor phosphorylation and cocaine-seeking behavior.Neuropsychopharmacology. 2013 Sep;38(10):1974-83. doi: 10.1038/npp.2013.96. Epub 2013 Apr 18. Neuropsychopharmacology. 2013. PMID: 23598433 Free PMC article.
-
Regulation of AMPA receptor trafficking in the nucleus accumbens by dopamine and cocaine.Neurotox Res. 2010 Nov;18(3-4):393-409. doi: 10.1007/s12640-010-9176-0. Epub 2010 Apr 2. Neurotox Res. 2010. PMID: 20361291 Free PMC article. Review.
Cited by
-
AMPA receptor plasticity in the nucleus accumbens after repeated exposure to cocaine.Neurosci Biobehav Rev. 2010 Nov;35(2):185-211. doi: 10.1016/j.neubiorev.2010.01.013. Epub 2010 Jan 28. Neurosci Biobehav Rev. 2010. PMID: 20109488 Free PMC article. Review.
-
The effect of CNQX on self-administration: present in nicotine, absent in methamphetamine model.Front Behav Neurosci. 2024 Jan 5;17:1305412. doi: 10.3389/fnbeh.2023.1305412. eCollection 2023. Front Behav Neurosci. 2024. PMID: 38249125 Free PMC article.
-
Disruption of GluA2 phosphorylation potentiates stress responsivity.Behav Brain Res. 2017 Aug 30;333:83-89. doi: 10.1016/j.bbr.2017.06.046. Epub 2017 Jun 28. Behav Brain Res. 2017. PMID: 28668281 Free PMC article.
-
Environmental novelty causes stress-like adaptations at nucleus accumbens synapses: implications for studying addiction-related plasticity.Neuropharmacology. 2011 Dec;61(7):1152-9. doi: 10.1016/j.neuropharm.2011.01.038. Epub 2011 Feb 16. Neuropharmacology. 2011. PMID: 21315090 Free PMC article.
-
Single Exposure to Cocaine Impairs Reinforcement Learning by Potentiating the Activity of Neurons in the Direct Striatal Pathway in Mice.Neurosci Bull. 2021 Aug;37(8):1119-1134. doi: 10.1007/s12264-021-00687-8. Epub 2021 Apr 27. Neurosci Bull. 2021. PMID: 33905097 Free PMC article.
References
-
- Anderson SM, Famous KR, Sadri-Vakili G, Kumaresan V, Schmidt HD, Bass CE, Terwilliger EF, Cha JH, Pierce RC. CaMKII: a biochemical bridge linking accumbens dopamine and glutamate systems in cocaine seeking. Nat Neurosci. 2008;11:344–353. - PubMed
-
- Bellone C, Lüscher C. Cocaine triggered AMPA receptor redistribution is reversed in vivo by mGluR-dependent long-term depression. Nat Neurosci. 2006;5:636–641. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
