Ghrelin in the human myometrium
- PMID: 20509935
- PMCID: PMC2887880
- DOI: 10.1186/1477-7827-8-55
Ghrelin in the human myometrium
Abstract
Background: Ghrelin is a 28-amino acid octanolyated peptide, synthesised primarily in the stomach. It stimulates growth hormone release, food intake and exhibits many other diverse effects. Our group have previously determined that ghrelin inhibited human contractility in vitro. The aim of this study therefore, was to investigate the expression of ghrelin, its receptor, the growth hormone secretagogue receptor type 1 (GHS-R1), ghrelin O-acyltransferase (GOAT) which catalyses ghrelin octanoylation, prohormone convertase 1/3 (PC1/3) responsible for pro-ghrelin processing, in human myometrium, during pregnancy prior to labour, during labour and in the non-pregnant state. Modulation of ghrelin and ghrelin receptor expression in cultured myometrial cells was also investigated.
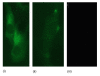
Methods: mRNA and protein were isolated from human myometrium and the myometrial smooth muscle cell line hTERT-HM; and real-time fluorescence RT-PCR, western blotting and fluorescence microscopy performed. The effects of beta-Estradiol and bacterial lipopolysaccharide (LPS) on hTERT-HM gene expression were evaluated by western blotting.
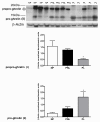
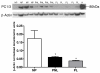
Results: We have reported for the first time the expression and processing of ghrelin, GHS-R1, GOAT and PC1/3 expression in human myometrium, and also the down-regulation of ghrelin mRNA and protein expression during labour. Furthermore, GHS-R1 protein expression significantly decreased at labour. Myometrial GOAT expression significantly increased during term non-labouring pregnancy in comparison to both non-pregnant and labouring myometrium. Mature PC1/3 protein expression was significantly decreased at term pregnancy and labour in comparison to non-pregnant myometrium. Ghrelin, GHS-R1, GOAT and PC1/3 mRNA and protein expression was also detected in the hTERT-HM cells. Ghrelin protein expression decreased upon LPS treatment in these cells while beta-Estradiol treatment increased GHS-R1 expression.
Conclusions: Ghrelin processing occurred in the human myometrium at term pregnancy and in the non-pregnant state. GOAT expression which increased during term non-labouring pregnancy demonstrating a similar expression pattern to prepro-ghrelin and GHS-R1, decreased at labour, signifying possible myometrial ghrelin acylation. Moreover, the presence of PC1/3 may contribute to pro-ghrelin processing. These results along with the previous in vitro data suggest that myometrially-produced and processed ghrelin plays a significant autocrine or paracrine role in the maintenance of relaxation in this tissue during pregnancy. Furthermore, the significant uterine modulators LPS and beta-Estradiol are involved in the regulation of ghrelin and ghrelin receptor expression respectively, in the human myometrium.
Figures










Similar articles
-
Expression of RHOGTPase regulators in human myometrium.Reprod Biol Endocrinol. 2008 Jan 11;6:1. doi: 10.1186/1477-7827-6-1. Reprod Biol Endocrinol. 2008. PMID: 18190708 Free PMC article.
-
Identification of a novel corticotropin-releasing hormone type 1beta-like receptor variant lacking Exon 13 in human pregnant myometrium regulated by estradiol-17beta and progesterone.Endocrinology. 2010 Oct;151(10):4959-68. doi: 10.1210/en.2010-0622. Epub 2010 Aug 11. Endocrinology. 2010. PMID: 20702571
-
Levels of corticotrophin-releasing hormone receptor subtype 1 mRNA in pregnancy and during labour in human myometrium measured by quantitative competitive PCR.J Mol Endocrinol. 1998 Oct;21(2):201-8. doi: 10.1677/jme.0.0210201. J Mol Endocrinol. 1998. PMID: 9801463
-
Integration of endocrine and mechanical signals in the regulation of myometrial functions during pregnancy and labour.Eur J Obstet Gynecol Reprod Biol. 2009 May;144 Suppl 1:S2-10. doi: 10.1016/j.ejogrb.2009.02.044. Epub 2009 Mar 18. Eur J Obstet Gynecol Reprod Biol. 2009. PMID: 19299064 Review.
-
Ghrelin gene products and the regulation of food intake and gut motility.Pharmacol Rev. 2009 Dec;61(4):430-81. doi: 10.1124/pr.109.001958. Pharmacol Rev. 2009. PMID: 20038570 Review.
Cited by
-
Decreased myometrial p160 ROCK-1 expression in obese women at term pregnancy.Reprod Biol Endocrinol. 2013 Aug 15;11:79. doi: 10.1186/1477-7827-11-79. Reprod Biol Endocrinol. 2013. PMID: 23948067 Free PMC article.
-
An overview of pharmacological approaches for management and repair of spinal cord injuries.Iran J Psychiatry. 2010 Fall;5(4):119-27. Iran J Psychiatry. 2010. PMID: 22952505 Free PMC article.
-
The Role of the Gastric Hormones Ghrelin and Nesfatin-1 in Reproduction.Int J Mol Sci. 2021 Oct 14;22(20):11059. doi: 10.3390/ijms222011059. Int J Mol Sci. 2021. PMID: 34681721 Free PMC article. Review.
-
Risk of Cesarean Delivery for Women with Obesity Using a Standardized Labor Induction Protocol.Am J Perinatol. 2021 Dec;38(14):1453-1458. doi: 10.1055/s-0041-1732459. Epub 2021 Jul 19. Am J Perinatol. 2021. PMID: 34282575 Free PMC article.
-
Vascular biology of uterine fibroids: connecting fibroids and vascular disorders.Reproduction. 2021 Jul 8;162(2):R1-R18. doi: 10.1530/REP-21-0087. Reproduction. 2021. PMID: 34034234 Free PMC article. Review.
References
-
- Gnanapavan S, Kola B, Bustin SA, Morris DG, McGee P, Fairclough P, Bhattacharya S, Carpenter R, Grossman AB, Korbonits M. The tissue distribution of the mRNA of ghrelin and subtypes of its receptor, GHS-R, in humans. J Clin Endocrinol Metab. 2002;87(6):2988. doi: 10.1210/jc.87.6.2988. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

