Estradiol enhances cell-associated paraoxonase 1 (PON1) activity in vitro without altering PON1 expression
- PMID: 20510879
- PMCID: PMC2900384
- DOI: 10.1016/j.bbrc.2010.05.120
Estradiol enhances cell-associated paraoxonase 1 (PON1) activity in vitro without altering PON1 expression
Abstract
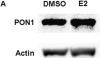
PON1 is a high density lipoprotein-associated enzyme that plays an important role in organophosphate detoxification and prevention of atherosclerosis. In vivo animal and human studies have indicated that estradiol (E2) supplementation enhances serum PON1 activity. In this study, we sought to determine if E2 directly up-regulates cell-associated PON1 activity in vitro and to characterize the mechanism of regulation. In vitro E2 treatment of both the human hepatoma cell line Huh7 and normal rat hepatocytes resulted in a 2- to 3-fold increase in cell-associated PON1 catalytic activity. E2 potently induced PON1 activity with average EC(50) values of 15nM for normal hepatocytes and 68nM for Huh7. The enhancement of PON1 activity by E2 was blocked by the estrogen receptor (ER) antagonist ICI 182,780 indicating that E2 was acting through the ER. The up-regulation of PON1 activity by E2 did not involve enhancement of PON1 mRNA or protein levels and did not promote secretion of PON1. Thus, E2 can enhance cell-associated PON1 activity in vitro without altering PON1 gene expression or protein level. Our data suggest that E2 may regulate the specific activity and/or stability of cell surface PON1.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures




Similar articles
-
Genistein as a potential inducer of the anti-atherogenic enzyme paraoxonase-1: studies in cultured hepatocytes in vitro and in rat liver in vivo.J Cell Mol Med. 2012 Oct;16(10):2331-41. doi: 10.1111/j.1582-4934.2012.01542.x. J Cell Mol Med. 2012. PMID: 22304296 Free PMC article.
-
Role of JNK and c-Jun signaling pathway in regulation of human serum paraoxonase 1 gene transcription by berberine in human HepG2 cells.Eur J Pharmacol. 2011 Jan 15;650(2-3):519-25. doi: 10.1016/j.ejphar.2010.10.051. Epub 2010 Oct 31. Eur J Pharmacol. 2011. PMID: 21044622
-
Quercetin up-regulates paraoxonase 1 gene expression via sterol regulatory element binding protein 2 that translocates from the endoplasmic reticulum to the nucleus where it specifically interacts with sterol responsive element-like sequence in paraoxonase 1 promoter in HuH7 liver cells.Metabolism. 2010 Sep;59(9):1372-8. doi: 10.1016/j.metabol.2009.12.025. Metabolism. 2010. PMID: 20728021
-
Modulation of paraoxonase (PON1) activity.Biochem Pharmacol. 2005 Feb 15;69(4):541-50. doi: 10.1016/j.bcp.2004.08.027. Biochem Pharmacol. 2005. PMID: 15670573 Review.
-
A review of paraoxonase 1 properties and diagnostic applications.Pol J Vet Sci. 2016;19(1):225-32. doi: 10.1515/pjvs-2016-0028. Pol J Vet Sci. 2016. PMID: 27096809 Review.
Cited by
-
High-density lipoprotein cholesterol levels are associated with major adverse cardiovascular events in male but not female patients with hypertension.Clin Cardiol. 2021 May;44(5):723-730. doi: 10.1002/clc.23606. Epub 2021 Mar 30. Clin Cardiol. 2021. PMID: 33786851 Free PMC article.
-
Paraoxonase 2 (PON2) in the mouse central nervous system: a neuroprotective role?Toxicol Appl Pharmacol. 2011 Nov 1;256(3):369-78. doi: 10.1016/j.taap.2011.02.014. Epub 2011 Feb 23. Toxicol Appl Pharmacol. 2011. PMID: 21354197 Free PMC article.
-
Effects of aerobic training on serum paraoxonase activity and its relationship with PON1-192 phenotypes in women.J Sport Health Sci. 2016 Dec;5(4):462-468. doi: 10.1016/j.jshs.2015.01.010. Epub 2015 May 28. J Sport Health Sci. 2016. PMID: 30356554 Free PMC article.
-
Modulatory Effect of Lifestyle-Related, Environmental and Genetic Factors on Paraoxonase-1 Activity: A Review.Int J Environ Res Public Health. 2023 Feb 5;20(4):2813. doi: 10.3390/ijerph20042813. Int J Environ Res Public Health. 2023. PMID: 36833509 Free PMC article. Review.
-
Paraoxonase 1 serum activity in women: the effects of menopause, the C(-107)T polymorphism and food intake.Arch Endocrinol Metab. 2019 May-Jun;63(3):272-279. doi: 10.20945/2359-3997000000130. Epub 2019 Apr 25. Arch Endocrinol Metab. 2019. PMID: 31038597 Free PMC article.
References
-
- Soran H, Younis NN, Charlton-Menys V, Durrington P. Variation in paraoxonase-1 activity and atherosclerosis. Curr Opin Lipidol. 2009;20:265–274. - PubMed
-
- Bhattacharyya T, Nicholls SJ, Topol EJ, Zhang R, Yang X, Schmitt D, Fu X, Shao M, Brennan DM, Ellis SG, Brennan ML, Allayee H, Lusis AJ, Hazen SL. Relationship of paraoxonase 1 (PON1) gene polymorphisms and functional activity with systemic oxidative stress and cardiovascular risk. JAMA. 2008;299:1265–1276. - PMC - PubMed
-
- Aviram M, Hardak E, Vaya J, Mahmood S, Milo S, Hoffman A, Billicke S, Draganov D, Rosenblat M. Human serum paraoxonases (PON1) Q and R selectively decrease lipid peroxides in human coronary and carotid atherosclerotic lesions: PON1 esterase and peroxidase-like activities. Circulation. 2000;101:2510–2517. - PubMed
-
- Rosenblat M, Gaidukov L, Khersonsky O, Vaya J, Oren R, Tawfik DS, Aviram M. The catalytic histidine dyad of high density lipoprotein-associated serum paraoxonase-1 (PON1) is essential for PON1-mediated inhibition of low density lipoprotein oxidation and stimulation of macrophage cholesterol efflux. J Biol Chem. 2006;281:7657–7665. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

